Page 135 - Read Online
P. 135
Page 12 of 14 Shi et al. Energy Mater 2023;3:300036 https://dx.doi.org/10.20517/energymater.2023.27
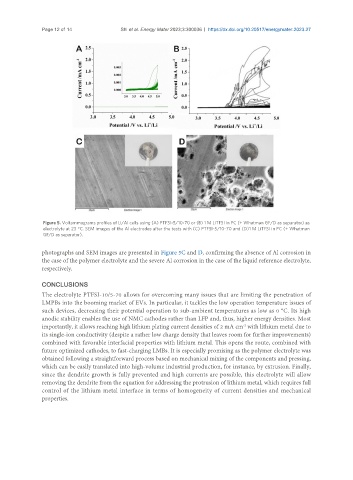
Figure 5. Voltammograms profiles of Li/Al cells using (A) PTFSI-5/10-70 or (B) 1 M LiTFSI in PC (+ Whatman GF/D as separator) as
electrolyte at 23 °C. SEM images of the Al electrodes after the tests with (C) PTFSI-5/10-70 and (D)1 M LiTFSI in PC (+ Whatman
GF/D as separator).
photographs and SEM images are presented in Figure 5C and D, confirming the absence of Al corrosion in
the case of the polymer electrolyte and the severe Al corrosion in the case of the liquid reference electrolyte,
respectively.
CONCLUSIONS
The electrolyte PTFSI-10/5-70 allows for overcoming many issues that are limiting the penetration of
LMPBs into the booming market of EVs. In particular, it tackles the low operation temperature issues of
such devices, decreasing their potential operation to sub-ambient temperatures as low as 0 °C. Its high
anodic stability enables the use of NMC cathodes rather than LFP and, thus, higher energy densities. Most
importantly, it allows reaching high lithium plating current densities of 2 mA cm with lithium metal due to
-2
its single-ion conductivity (despite a rather low charge density that leaves room for further improvements)
combined with favorable interfacial properties with lithium metal. This opens the route, combined with
future optimized cathodes, to fast-charging LMBs. It is especially promising as the polymer electrolyte was
obtained following a straightforward process based on mechanical mixing of the components and pressing,
which can be easily translated into high-volume industrial production, for instance, by extrusion. Finally,
since the dendrite growth is fully prevented and high currents are possible, this electrolyte will allow
removing the dendrite from the equation for addressing the protrusion of lithium metal, which requires full
control of the lithium metal interface in terms of homogeneity of current densities and mechanical
properties.