Page 140 - Read Online
P. 140
Shipitsyn et al. Energy Mater 2023;3:300038 https://dx.doi.org/10.20517/energymater.2023.22 Page 3 of 37
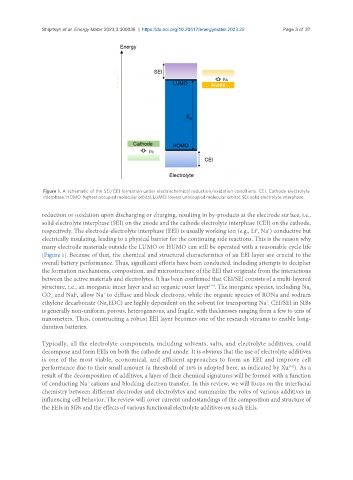
Figure 1. A schematic of the SEI/CEI formation under electrochemical reduction/oxidation conditions. CEI: Cathode electrolyte
interphase; HOMO: highest occupied molecular orbital; LUMO: lowest unoccupied molecular orbital; SEI: solid electrolyte interphase.
reduction or oxidation upon discharging or charging, resulting in by-products at the electrode surface, i.e.,
solid electrolyte interphase (SEI) on the anode and the cathode electrolyte interphase (CEI) on the cathode,
respectively. The electrode-electrolyte interphase (EEI) is usually working ion (e.g., Li , Na ) conductive but
+
+
electrically insulating, leading to a physical barrier for the continuing side reactions. This is the reason why
many electrode materials outside the LUMO or HUMO can still be operated with a reasonable cycle life
[Figure 1]. Because of that, the chemical and structural characteristics of an EEI layer are crucial to the
overall battery performance. Thus, significant efforts have been conducted, including attempts to decipher
the formation mechanisms, composition, and microstructure of the EEI that originate from the interactions
between the active materials and electrolytes. It has been confirmed that CEI/SEI consists of a multi-layered
structure, i.e., an inorganic inner layer and an organic outer layer . The inorganic species, including Na 2
[10]
CO and NaF, allow Na to diffuse and block electrons, while the organic species of RONa and sodium
+
3
ethylene decarbonate (Na EDC) are highly dependent on the solvent for transporting Na . CEI/SEI in SIBs
+
2
is generally non-uniform, porous, heterogeneous, and fragile, with thicknesses ranging from a few to tens of
nanometers. Thus, constructing a robust EEI layer becomes one of the research streams to enable long-
duration batteries.
Typically, all the electrolyte components, including solvents, salts, and electrolyte additives, could
decompose and form EEIs on both the cathode and anode. It is obvious that the use of electrolyte additives
is one of the most viable, economical, and efficient approaches to form an EEI and improve cell
performance due to their small amount (a threshold of 10% is adopted here, as indicated by Xu ). As a
[16]
result of the decomposition of additives, a layer of their chemical signatures will be formed with a function
+
of conducting Na cations and blocking electron transfer. In this review, we will focus on the interfacial
chemistry between different electrodes and electrolytes and summarize the roles of various additives in
influencing cell behavior. The review will cover current understandings of the composition and structure of
the EEIs in SIBs and the effects of various functional electrolyte additives on such EEIs.