Page 134 - Read Online
P. 134
Shi et al. Energy Mater 2023;3:300036 https://dx.doi.org/10.20517/energymater.2023.27 Page 11 of 14
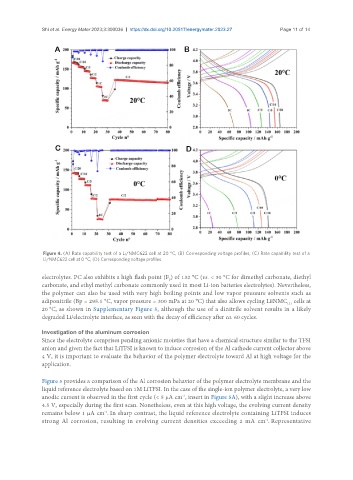
Figure 4. (A) Rate capability test of a Li/NMC622 cell at 20 °C, (B) Corresponding voltage profiles, (C) Rate capability test of a
Li/NMC622 cell at 0 °C, (D) Corresponding voltage profiles.
electrolytes. PC also exhibits a high flash point (F ) of 132 °C (vs. < 30 °C for dimethyl carbonate, diethyl
p
carbonate, and ethyl methyl carbonate commonly used in most Li-ion batteries electrolytes). Nevertheless,
the polymer can also be used with very high boiling points and low vapor pressure solvents such as
adiponitrile (Bp = 295.1 °C, vapor pressure = 300 mPa at 20 °C) that also allows cycling LiǁNMC cells at
111
20 °C, as shown in Supplementary Figure 5, although the use of a dinitrile solvent results in a likely
degraded Li/electrolyte interface, as seen with the decay of efficiency after ca. 60 cycles.
Investigation of the aluminum corrosion
Since the electrolyte comprises pending anionic moieties that have a chemical structure similar to the TFSI
anion and given the fact that LiTFSI is known to induce corrosion of the Al cathode current collector above
4 V, it is important to evaluate the behavior of the polymer electrolyte toward Al at high voltage for the
application.
Figure 5 provides a comparison of the Al corrosion behavior of the polymer electrolyte membrane and the
liquid reference electrolyte based on 1M LiTFSI. In the case of the single-ion polymer electrolyte, a very low
anodic current is observed in the first cycle (< 5 µA cm , insert in Figure 5A), with a slight increase above
-2
4.5 V, especially during the first scan. Nonetheless, even at this high voltage, the evolving current density
remains below 1 µA cm . In sharp contrast, the liquid reference electrolyte containing LiTFSI induces
-2
-2
strong Al corrosion, resulting in evolving current densities exceeding 2 mA cm . Representative