Page 133 - Read Online
P. 133
Page 10 of 14 Shi et al. Energy Mater 2023;3:300036 https://dx.doi.org/10.20517/energymater.2023.27
-2
-2
-2
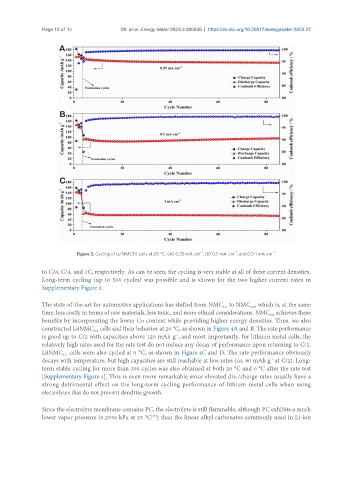
Figure 3. Cycling of Li/NMC111 cells at 20 °C. (A) 0.25 mA cm , (B) 0.5 mA cm , and (C) 1 mA cm .
to C/8, C/4, and 1C, respectively. As can be seen, the cycling is very stable at all of these current densities.
Long-term cycling (up to 500 cycles) was possible and is shown for the two higher current rates in
Supplementary Figure 3.
The state-of-the-art for automotive applications has shifted from NMC to NMC , which is, at the same
111
622
time, less costly in terms of raw materials, less toxic, and more ethical considerations. NMC achieves these
622
benefits by incorporating the lower Co content while providing higher energy densities. Thus, we also
constructed LiǁNMC cells and their behavior at 20 °C, as shown in Figure 4A and B. The rate performance
622
is good up to C/2 with capacities above 120 mAh g , and most importantly, for lithium metal cells, the
-1
relatively high rates used for the rate test do not induce any decay of performance upon returning to C/2.
LiǁNMC cells were also cycled at 0 °C, as shown in Figure 4C and D. The rate performance obviously
622
decays with temperature, but high capacities are still reachable at low rates (ca. 80 mAh g at C/2). Long-
-1
term stable cycling for more than 300 cycles was also obtained at both 20 °C and 0 °C after the rate test
[Supplementary Figure 4]. This is even more remarkable since elevated dis-/charge rates usually have a
strong detrimental effect on the long-term cycling performance of lithium metal cells when using
electrolytes that do not prevent dendrite growth.
Since the electrolyte membrane contains PC, the electrolyte is still flammable, although PC exhibits a much
[36]
lower vapor pressure (0.2936 kPa at 25 °C ) than the linear alkyl carbonates commonly used in Li-ion