Page 50 - Read Online
P. 50
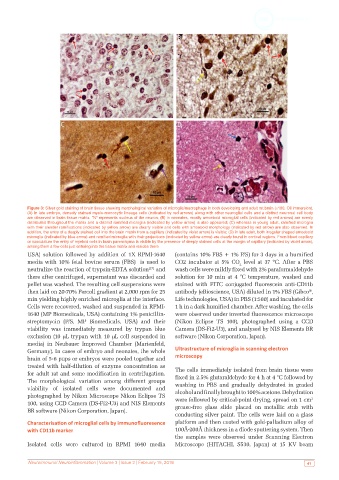
Figure 3: Silver gold staining of brain tissue showing morphological variation of microglia/macrophage in both developing and adult rat brain (×100, Oil immersion).
(A) In late embryo, densely stained myelo-monocytic lineage cells (indicated by red arrows) along with other neuroglial cells and a distinct neuronal cell body
are observed in brain tissue matrix. “N” represents nucleus of the neuron; (B) in neonates, mostly amoeboid microglial cells (indicated by red arrows) are evenly
distributed throughout the matrix and a distinct ramified microglia (indicated by yellow arrow) is also appeared; (C) whereas in young adult, ramified microglia
with their slender ramifications (indicated by yellow arrow) are clearly visible and cells with amoeboid morphology (indicated by red arrow) are also observed. In
addition, the entry of a deeply stained cell into the brain matrix from a capillary (indicated by violet arrow) is visible; (D) in late adult, both irregular shaped amoeboid
microglia (indicated by blue arrow) and ramified microglia with their projections (indicated by yellow arrow) are clearly found in cortical regions. From blood capillary
or vasculature the entry of myeloid cells in brain parenchyma is visible by the presence of deeply stained cells at the margin of capillary (indicated by violet arrow),
among them a few cells just entering into the tissue matrix and resides there
USA) solution followed by addition of 1X RPMI-1640 (contains 10% FBS + 1% P/S) for 3 days in a humified
media with 10% fetal bovine serum (FBS) is used to CO2 incubator at 5% CO level at 37 ºC. After a PBS
2
neutralize the reaction of trypsin-EDTA solution and wash cells were mildly fixed with 2% paraformaldehyde
[27]
there after centrifuged, supernatant was discarded and solution for 10 min at 4 °C temperature, washed and
pellet was washed. The resulting cell suspensions were stained with FITC conjugated fluorescein anti-CD11b
then laid on 20-70% Percoll gradient at 2,000 rpm for 25 antibody (eBioscience, USA) diluted in 1% FBS (Gibco ,
®
min yielding highly enriched microglia at the interface. Life technologies, USA) in PBS (1:500) and incubated for
Cells were recovered, washed and suspended in RPMI- 1 h in a dark humified chamber. After washing, the cells
1640 (MP Biomedicals, USA) containing 1% penicillin- were observed under inverted fluorescence microscope
streptomycin (P/S, MP Biomedicals, USA) and their (Nikon Eclipse TS 100), photographed using a CCD
viability was immediately measured by trypan blue Camera (DS-Fi2-U3), and analysed by NIS Elements BR
exclusion (10 µL trypan with 10 µL cell suspended in software (Nikon Corporation, Japan).
media) in Neubauer Improved Chamber (Marienfeld,
Germany). In cases of embryo and neonates, the whole Ultrastructure of microglia in scanning electron
brain of 5-6 pups or embryos were pooled together and microscopy
treated with half-dilution of enzyme concentration as
for adult rat and some modification in centrifugation. The cells immediately isolated from brain tissue were
The morphological variation among different groups fixed in 2.5% glutaraldehyde for 4 h at 4 °C followed by
viability of isolated cells were documented and washing in PBS and gradually dehydrated in graded
photographed by Nikon Microscope Nikon Eclipse TS alcohol and finally brought to 100% acetone. Dehydration
2
100, using CCD Camera (DS-Fi2-U3) and NIS Elements were followed by critical-point drying, spread on 1 cm
grease-free glass slide placed on metallic stub with
BR software (Nikon Corporation, Japan).
conducting silver paint. The cells were laid on a glass
Characterisation of microglial cells by immunofluoresence platform and then coated with gold-palladium alloy of
with CD11b marker 100Å-200Å thickness in a diode sputtering system. Then
the samples were observed under Scanning Electron
Isolated cells were cultured in RPMI 1640 media Microscope (HITACHI, S530, Japan) at 15 KV beam
Neuroimmunol Neuroinflammation | Volume 3 | Issue 2 | February 15, 2016 41