Page 81 - Read Online
P. 81
Page 16 of 28 Choi et al. Energy Mater. 2025, 5, 500106 https://dx.doi.org/10.20517/energymater.2025.50
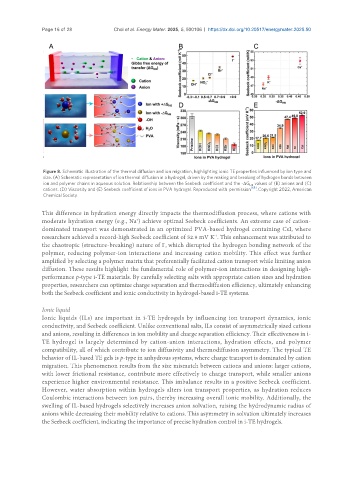
Figure 8. Schematic illustration of the thermal diffusion and ion migration, highlighting ionic TE properties influenced by ion type and
size. (A) Schematic representation of ion thermal diffusion in a hydrogel, driven by the making and breaking of hydrogen bonds between
ion and polymer chains in aqueous solution. Relationship between the Seebeck coefficient and the -ΔG values of (B) anions and (C)
HB
[68]
cations. (D) Viscosity and (E) Seebeck coefficient of ions in PVA hydrogel. Reproduced with permission . Copyright 2022, American
Chemical Society.
This difference in hydration energy directly impacts the thermodiffusion process, where cations with
+
moderate hydration energy (e.g., Na ) achieve optimal Seebeck coefficients. An extreme case of cation-
dominated transport was demonstrated in an optimized PVA-based hydrogel containing CsI, where
researchers achieved a record-high Seebeck coefficient of 52.9 mV K . This enhancement was attributed to
-1
the chaotropic (structure-breaking) nature of I , which disrupted the hydrogen bonding network of the
-
polymer, reducing polymer-ion interactions and increasing cation mobility. This effect was further
amplified by selecting a polymer matrix that preferentially facilitated cation transport while limiting anion
diffusion. These results highlight the fundamental role of polymer-ion interactions in designing high-
performance p-type i-TE materials. By carefully selecting salts with appropriate cation sizes and hydration
properties, researchers can optimize charge separation and thermodiffusion efficiency, ultimately enhancing
both the Seebeck coefficient and ionic conductivity in hydrogel-based i-TE systems.
Ionic liquid
Ionic liquids (ILs) are important in i-TE hydrogels by influencing ion transport dynamics, ionic
conductivity, and Seebeck coefficient. Unlike conventional salts, ILs consist of asymmetrically sized cations
and anions, resulting in differences in ion mobility and charge separation efficiency. Their effectiveness in i-
TE hydrogel is largely determined by cation-anion interactions, hydration effects, and polymer
compatibility, all of which contribute to ion diffusivity and thermodiffusion asymmetry. The typical TE
behavior of IL-based TE gels is p-type in anhydrous systems, where charge transport is dominated by cation
migration. This phenomenon results from the size mismatch between cations and anions: larger cations,
with lower frictional resistance, contribute more effectively to charge transport, while smaller anions
experience higher environmental resistance. This imbalance results in a positive Seebeck coefficient.
However, water absorption within hydrogels alters ion transport properties, as hydration reduces
Coulombic interactions between ion pairs, thereby increasing overall ionic mobility. Additionally, the
swelling of IL-based hydrogels selectively increases anion solvation, raising the hydrodynamic radius of
anions while decreasing their mobility relative to cations. This asymmetry in solvation ultimately increases
the Seebeck coefficient, indicating the importance of precise hydration control in i-TE hydrogels.