Page 76 - Read Online
P. 76
Choi et al. Energy Mater. 2025, 5, 500106 https://dx.doi.org/10.20517/energymater.2025.50 Page 11 of 28
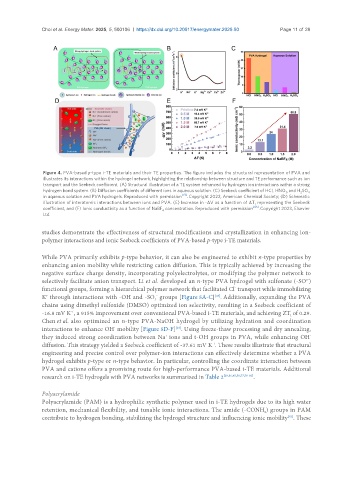
Figure 4. PVA-based p-type i-TE materials and their TE properties. The figure includes the structural representation of PVA and
illustrates its interactions within the hydrogel network, highlighting the relationship between structure and TE performance such as ion
transport and the Seebeck coefficient. (A) Structural illustration of a TE system enhanced by hydrogen ion interactions within a strong
hydrogen bond system. (B) Diffusion coefficients of different ions in aqueous solution. (C) Seebeck coefficient of HCl, HNO , and H SO
3 2 4
[79]
in aqueous solution and PVA hydrogels. Reproduced with permission . Copyright 2022, American Chemical Society; (D) Schematic
illustration of interatomic interactions between ions and PVA. (E) Increase in -ΔV as a function of ΔT, representing the Seebeck
coefficient, and (F) Ionic conductivity as a function of NaBF concentration. Reproduced with permission [45] . Copyright 2023, Elsevier
4
Ltd.
studies demonstrate the effectiveness of structural modifications and crystallization in enhancing ion-
polymer interactions and ionic Seebeck coefficients of PVA-based p-type i-TE materials.
While PVA primarily exhibits p-type behavior, it can also be engineered to exhibit n-type properties by
enhancing anion mobility while restricting cation diffusion. This is typically achieved by increasing the
negative surface charge density, incorporating polyelectrolytes, or modifying the polymer network to
selectively facilitate anion transport. Li et al. developed an n-type PVA hydrogel with sulfonate (-SO )
3-
functional groups, forming a hierarchical polymer network that facilitated Cl transport while immobilizing
-
+
K through interactions with -OH and -SO groups [Figure 5A-C] . Additionally, expanding the PVA
-
[36]
3
chains using dimethyl sulfoxide (DMSO) optimized ion selectivity, resulting in a Seebeck coefficient of
-16.8 mV K , a 915% improvement over conventional PVA-based i-TE materials, and achieving ZT of 0.29.
-1
i
Chen et al. also optimized an n-type PVA-NaOH hydrogel by utilizing hydration and coordination
interactions to enhance OH mobility [Figure 5D-F] . Using freeze-thaw processing and dry annealing,
[29]
-
+
-
they induced strong coordination between Na ions and t-OH groups in PVA, while enhancing OH
diffusion. This strategy yielded a Seebeck coefficient of -37.61 mV K . These results illustrate that structural
-1
engineering and precise control over polymer-ion interactions can effectively determine whether a PVA
hydrogel exhibits p-type or n-type behavior. In particular, controlling the coordinate interaction between
PVA and cations offers a promising route for high-performance PVA-based i-TE materials. Additional
research on i-TE hydrogels with PVA networks is summarized in Table 2 [29,36,45,50,77,79-81] .
Polyacrylamide
Polyacrylamide (PAM) is a hydrophilic synthetic polymer used in i-TE hydrogels due to its high water
retention, mechanical flexibility, and tunable ionic interactions. The amide (-CONH ) groups in PAM
2
contribute to hydrogen bonding, stabilizing the hydrogel structure and influencing ionic mobility . These
[82]