Page 77 - Read Online
P. 77
Page 12 of 28 Choi et al. Energy Mater. 2025, 5, 500106 https://dx.doi.org/10.20517/energymater.2025.50
Table 2. TE performance of i-TE hydrogels with PVA networks
Authors Polymer Ion conductor Type S [mV/K] σ [mS/cm] κ [W/mK]
i
i
i
Hsiao et al. [45] PVA Na[BF ] p 66.7 31.4 -
4
PEG
[79]
Chen et al. PVA HCl p 38.2 1.89 0.46
[80]
Horike et al. PVA [Rmim]Cl p 10 0.16 0.29
[29]
Chen et al. PVA NaOH n -37.6 0.00736 0.42
Li et al. [36] PVA KCl n -16.8 17.7 0.5
[50] 2−
Chen et al. PVA cellulose [CuCl ] n -26.3 8.47 0.47
4
Chen et al. [77] PVA NaOH n -20.7 0.62 -
BC
Hu et al. [81] PVA ZnSO 4 n -3.69 0.15 -
PVP
S : Ionic Seebeck coefficient; σ : ionic conductivity; κ : thermal conductivity; PEG: polyethylene glycol; PVP: polyvinylpyrrolidone; BC: bacterial
i
i
i
cellulose.
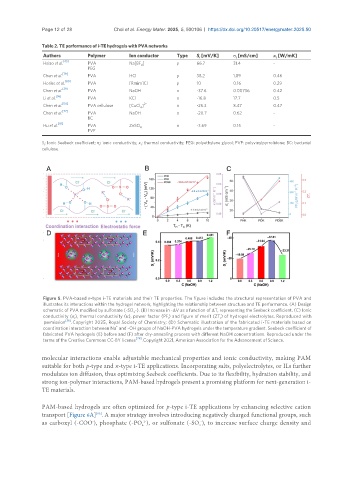
Figure 5. PVA-based n-type i-TE materials and their TE properties. The figure includes the structural representation of PVA and
illustrates its interactions within the hydrogel network, highlighting the relationship between structure and TE performance. (A) Design
schematic of PVA modified by sulfonate (-SO -). (B) Increase in -ΔV as a function of ΔT, representing the Seebeck coefficient. (C) Ionic
3
conductivity (σ), thermal conductivity (κ), power factor (PF) and figure of merit (ZT) of hydrogel electrolytes. Reproduced with
i
i
i
permission [36] . Copyright 2025, Royal Society of Chemistry; (D) Schematic illustration of the fabricated i-TE materials based on
+
coordination interaction between Na and -OH groups of NaOH-PVA hydrogels under the temperature gradient. Seebeck coefficient of
fabricated PVA hydrogels (E) before and (F) after dry-annealing process with different NaOH concentrations. Reproduced under the
terms of the Creative Commons CC-BY license [29] . Copyright 2021, American Association for the Advancement of Science.
molecular interactions enable adjustable mechanical properties and ionic conductivity, making PAM
suitable for both p-type and n-type i-TE applications. Incorporating salts, polyelectrolytes, or ILs further
modulates ion diffusion, thus optimizing Seebeck coefficients. Due to its flexibility, hydration stability, and
strong ion-polymer interactions, PAM-based hydrogels present a promising platform for next-generation i-
TE materials.
PAM-based hydrogels are often optimized for p-type i-TE applications by enhancing selective cation
[83]
transport [Figure 6A] . A major strategy involves introducing negatively charged functional groups, such
-
as carboxyl (-COO ), phosphate (-PO ), or sulfonate (-SO ), to increase surface charge density and
2-
-
3
4