Page 80 - Read Online
P. 80
Choi et al. Energy Mater. 2025, 5, 500106 https://dx.doi.org/10.20517/energymater.2025.50 Page 15 of 28
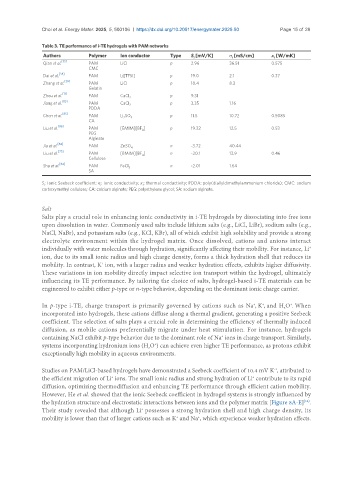
Table 3. TE performance of i-TE hydrogels with PAM networks
Authors Polymer Ion conductor Type S [mV/K] σ [mS/cm] κ [W/mK]
i
i
i
Qian et al. [35] PAM LiCl p 2.96 36.51 0.575
CMC
[38]
Dai et al. PAM Li[TFSI] p 19.0 2.1 0.37
[39]
Zhang et al. PAM LiCl p 10.4 8.3
Gelatin
Zhou et al. [71] PAM CaCl 2 p 9.31
[83]
Jiang et al. PAM CaCl 2 p 3.35 1.16
PDDA
[85]
Chen et al. PAM Li SO p 11.5 10.72 0.5085
2 4
CA
[86]
Liu et al. PAM [EMIM][BF ] p 19.32 12.5 0.53
4
PEG
Alginate
[64]
Jia et al. PAM ZnSO 4 n -3.72 40.44
[75]
Liu et al. PAM [BMIM][BF ] n -20.1 13.9 0.46
4
Cellulose
Sha et al. [84] PAM FeCl 3 n -2.01 1.64
SA
S : Ionic Seebeck coefficient; σ : ionic conductivity; κ : thermal conductivity; PDDA: poly(diallyldimethylammonium chloride); CMC: sodium
i i i
carboxymethyl cellulose; CA: calcium alginate; PEG: polyethylene glycol; SA: sodium alginate.
Salt
Salts play a crucial role in enhancing ionic conductivity in i-TE hydrogels by dissociating into free ions
upon dissolution in water. Commonly used salts include lithium salts (e.g., LiCl, LiBr), sodium salts (e.g.,
NaCl, NaBr), and potassium salts (e.g., KCl, KBr), all of which exhibit high solubility and provide a strong
electrolyte environment within the hydrogel matrix. Once dissolved, cations and anions interact
individually with water molecules through hydration, significantly affecting their mobility. For instance, Li
+
ion, due to its small ionic radius and high charge density, forms a thick hydration shell that reduces its
mobility. In contrast, K ion, with a larger radius and weaker hydration effects, exhibits higher diffusivity.
+
These variations in ion mobility directly impact selective ion transport within the hydrogel, ultimately
influencing its TE performance. By tailoring the choice of salts, hydrogel-based i-TE materials can be
engineered to exhibit either p-type or n-type behavior, depending on the dominant ionic charge carrier.
+
+
In p-type i-TE, charge transport is primarily governed by cations such as Na , K , and H O . When
+
3
incorporated into hydrogels, these cations diffuse along a thermal gradient, generating a positive Seebeck
coefficient. The selection of salts plays a crucial role in determining the efficiency of thermally induced
diffusion, as mobile cations preferentially migrate under heat stimulation. For instance, hydrogels
+
containing NaCl exhibit p-type behavior due to the dominant role of Na ions in charge transport. Similarly,
systems incorporating hydronium ions (H O ) can achieve even higher TE performance, as protons exhibit
+
3
exceptionally high mobility in aqueous environments.
Studies on PAM/LiCl-based hydrogels have demonstrated a Seebeck coefficient of 10.4 mV K , attributed to
-1
the efficient migration of Li ions. The small ionic radius and strong hydration of Li contribute to its rapid
+
+
diffusion, optimizing thermodiffusion and enhancing TE performance through efficient cation mobility.
However, He et al. showed that the ionic Seebeck coefficient in hydrogel systems is strongly influenced by
the hydration structure and electrostatic interactions between ions and the polymer matrix [Figure 8A-E] .
[68]
Their study revealed that although Li possesses a strong hydration shell and high charge density, its
+
+
mobility is lower than that of larger cations such as K and Na , which experience weaker hydration effects.
+