Page 78 - Read Online
P. 78
Choi et al. Energy Mater. 2025, 5, 500106 https://dx.doi.org/10.20517/energymater.2025.50 Page 13 of 28
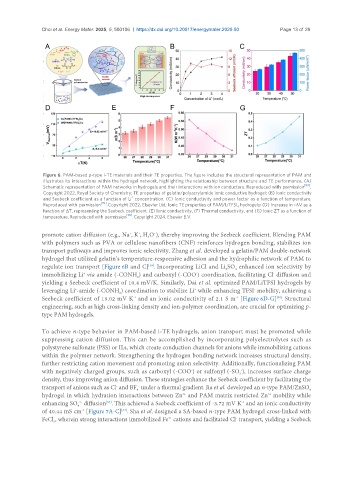
Figure 6. PAM-based p-type i-TE materials and their TE properties. The figure includes the structural representation of PAM and
illustrates its interactions within the hydrogel network, highlighting the relationship between structure and TE performance. (A)
Schematic representation of PAM networks in hydrogels and their interactions with ion conductors. Reproduced with permission [83] .
Copyright 2022, Royal Society of Chemistry; TE properties of gelatin/polyacrylamide ionic conductive hydrogel: (B) Ionic conductivity
+
and Seebeck coefficient as a function of Li concentration. (C) Ionic conductivity and power factor as a function of temperature.
[39]
Reproduced with permission . Copyright 2022, Elsevier Ltd; Ionic TE properties of PAM/LiTFSI hydrogels: (D) Increase in -ΔV as a
4
function of ΔT, representing the Seebeck coefficient. (E) Ionic conductivity, (F) Thermal conductivity, and (G) Ionic ZT as a function of
temperature. Reproduced with permission [38] . Copyright 2024, Elsevier B.V.
promote cation diffusion (e.g., Na , K , H O ), thereby improving the Seebeck coefficient. Blending PAM
+
+
+
3
with polymers such as PVA or cellulose nanofibers (CNF) reinforces hydrogen bonding, stabilizes ion
transport pathways and improves ionic selectivity. Zhang et al. developed a gelatin/PAM double-network
hydrogel that utilized gelatin’s temperature-responsive adhesion and the hydrophilic network of PAM to
regulate ion transport [Figure 6B and C] . Incorporating LiCl and Li SO enhanced ion selectivity by
[39]
4
2
immobilizing Li via amide (-CONH ) and carboxyl (-COO ) coordination, facilitating Cl diffusion and
-
-
+
2
yielding a Seebeck coefficient of 10.4 mV/K. Similarly, Dai et al. optimized PAM/LiTFSI hydrogels by
+
-
+
leveraging Li -amide (-CONH ) coordination to stabilize Li while enhancing TFSI mobility, achieving a
2
-1
[38]
-1
Seebeck coefficient of 19.02 mV K and an ionic conductivity of 2.1 S m [Figure 6D-G] . Structural
engineering, such as high cross-linking density and ion-polymer coordination, are crucial for optimizing p-
type PAM hydrogels.
To achieve n-type behavior in PAM-based i-TE hydrogels, anion transport must be promoted while
suppressing cation diffusion. This can be accomplished by incorporating polyelectrolytes such as
polystyrene sulfonate (PSS) or ILs, which create conduction channels for anions while immobilizing cations
within the polymer network. Strengthening the hydrogen bonding network increases structural density,
further restricting cation movement and promoting anion selectivity. Additionally, functionalizing PAM
with negatively charged groups, such as carboxyl (-COO ) or sulfonyl (-SO ), increases surface charge
-
-
3
density, thus improving anion diffusion. These strategies enhance the Seebeck coefficient by facilitating the
transport of anions such as Cl and BF under a thermal gradient. Jia et al. developed an n-type PAM/ZnSO
-
-
4
4
2+
2+
hydrogel in which hydration interactions between Zn and PAM matrix restricted Zn mobility while
enhancing SO diffusion . This achieved a Seebeck coefficient of -3.72 mV K and an ionic conductivity
-1
2-
[64]
4
of 40.44 mS cm [Figure 7A-C] . Sha et al. designed a SA-based n-type PAM hydrogel cross-linked with
-1
[64]
FeCl , wherein strong interactions immobilized Fe cations and facilitated Cl transport, yielding a Seebeck
-
3+
3