Page 79 - Read Online
P. 79
Page 14 of 28 Choi et al. Energy Mater. 2025, 5, 500106 https://dx.doi.org/10.20517/energymater.2025.50
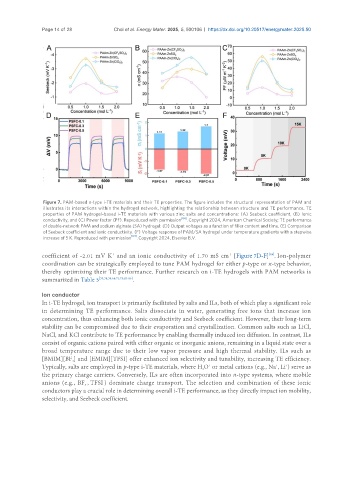
Figure 7. PAM-based n-type i-TE materials and their TE properties. The figure includes the structural representation of PAM and
illustrates its interactions within the hydrogel network, highlighting the relationship between structure and TE performance. TE
properties of PAM hydrogel-based i-TE materials with various zinc salts and concentrations: (A) Seebeck coefficient, (B) Ionic
conductivity, and (C) Power factor (PF). Reproduced with permission [64] . Copyright 2024, American Chemical Society; TE performance
of double-network PAM and sodium alginate (SA) hydrogel: (D) Output voltages as a function of filler content and time. (E) Comparison
of Seebeck coefficient and ionic conductivity. (F) Voltage response of PAM/SA hydrogel under temperature gradients with a stepwise
increase of 5 K. Reproduced with permission [84] . Copyright 2024, Elsevier B.V.
coefficient of -2.01 mV K and an ionic conductivity of 1.70 mS cm [Figure 7D-F] . Ion-polymer
-1
-1
[84]
coordination can be strategically employed to tune PAM hydrogel for either p-type or n-type behavior,
thereby optimizing their TE performance. Further research on i-TE hydrogels with PAM networks is
summarized in Table 3 [35,38,39,64,71,75,83-86] .
Ion conductor
In i-TE hydrogel, ion transport is primarily facilitated by salts and ILs, both of which play a significant role
in determining TE performance. Salts dissociate in water, generating free ions that increase ion
concentration, thus enhancing both ionic conductivity and Seebeck coefficient. However, their long-term
stability can be compromised due to their evaporation and crystallization. Common salts such as LiCl,
NaCl, and KCl contribute to TE performance by enabling thermally induced ion diffusion. In contrast, ILs
consist of organic cations paired with either organic or inorganic anions, remaining in a liquid state over a
broad temperature range due to their low vapor pressure and high thermal stability. ILs such as
[BMIM][BF ] and [EMIM][TFSI] offer enhanced ion selectivity and tunability, increasing TE efficiency.
4
+
+
Typically, salts are employed in p-type i-TE materials, where H O or metal cations (e.g., Na , Li ) serve as
+
3
the primary charge carriers. Conversely, ILs are often incorporated into n-type systems, where mobile
-
anions (e.g., BF , TFSI ) dominate charge transport. The selection and combination of these ionic
-
4
conductors play a crucial role in determining overall i-TE performance, as they directly impact ion mobility,
selectivity, and Seebeck coefficient.