Page 161 - Read Online
P. 161
Shipitsyn et al. Energy Mater 2023;3:300038 https://dx.doi.org/10.20517/energymater.2023.22 Page 23 of 37
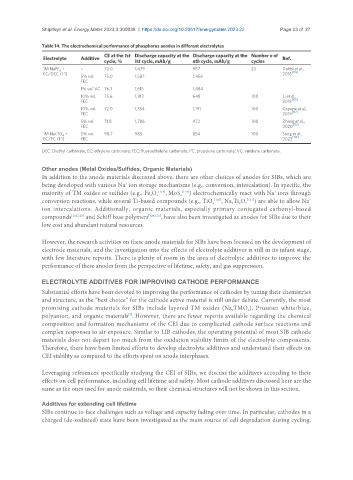
Table 14. The electrochemical performance of phosphorus anodes in different electrolytes
CE at the 1st Discharge capacity at the Discharge capacity at the Number n of
Electrolyte Additive Ref.
cycle, % 1st cycle, mAh/g nth cycle, mAh/g cycles
1M NaPF + - 72.0 1,479 957 23 Dahbi et al.,
6 [114]
EC/DEC (1:1) 2016
5% vol. 75.0 1,587 1,458
FEC
1% vol. VC 76.3 1,615 1,484
10% vol. 75.6 1,183 648 100 Li et al.,
[115]
FEC 2018
10% vol. 72.0 1,354 1,191 100 Capone et al.,
FEC 2019 [117]
5% vol. 71.8 1,786 972 100 Zhang et al.,
[112]
FEC 2020
1M NaClO + 5% vol. 98.7 985 854 100 Song et al.,
4
EC/PC (1:1) FEC 2023 [116]
DEC: Diethyl carbonate; EC: ethylene carbonate; FEC: fluoroethylene carbonate; PC: propylene carbonate; VC: vinylene carbonate.
Other anodes (Metal Oxides/Sulfides, Organic Materials)
In addition to the anode materials discussed above, there are other choices of anodes for SIBs, which are
being developed with various Na ion storage mechanisms (e.g., conversion, intercalation). In specific, the
+
majority of TM oxides or sulfides (e.g., Fe O 3 [118] , MoS 2 [119] ) electrochemically react with Na ions through
+
2
conversion reactions, while several Ti-based compounds (e.g., TiO 2 [120] , Na Ti O 7 [121] ) are able to allow Na
+
2
3
ion intercalations. Additionally, organic materials, especially primary conjugated carbonyl-based
compounds [122,123] and Schiff base polymers [124,125] , have also been investigated as anodes for SIBs due to their
low cost and abundant natural resources.
However, the research activities on these anode materials for SIBs have been focused on the development of
electrode materials, and the investigation into the effects of electrolyte additives is still in its infant stage,
with few literature reports. There is plenty of room in the area of electrolyte additives to improve the
performance of these anodes from the perspective of lifetime, safety, and gas suppression.
ELECTROLYTE ADDITIVES FOR IMPROVING CATHODE PERFORMANCE
Substantial efforts have been devoted to improving the performance of cathodes by tuning their chemistries
and structure, as the “best choice” for the cathode active material is still under debate. Currently, the most
promising cathode materials for SIBs include layered TM oxides (Na TMO ), Prussian white/blue,
x
2
polyanion, and organic materials . However, there are fewer reports available regarding the chemical
[7]
composition and formation mechanisms of the CEI due to complicated cathode surface reactions and
complex responses to air exposure. Similar to LIB cathodes, the operating potential of most SIB cathode
materials does not depart too much from the oxidation stability limits of the electrolyte components.
Therefore, there have been limited efforts to develop electrolyte additives and understand their effects on
CEI stability as compared to the efforts spent on anode interphases.
Leveraging references specifically studying the CEI of SIBs, we discuss the additives according to their
effects on cell performance, including cell lifetime and safety. Most cathode additives discussed here are the
same as the ones used for anode materials, so their chemical structures will not be shown in this section.
Additives for extending cell lifetime
SIBs continue to face challenges such as voltage and capacity fading over time. In particular, cathodes in a
charged (de-sodiated) state have been investigated as the main source of cell degradation during cycling,