Page 163 - Read Online
P. 163
Shipitsyn et al. Energy Mater 2023;3:300038 https://dx.doi.org/10.20517/energymater.2023.22 Page 25 of 37
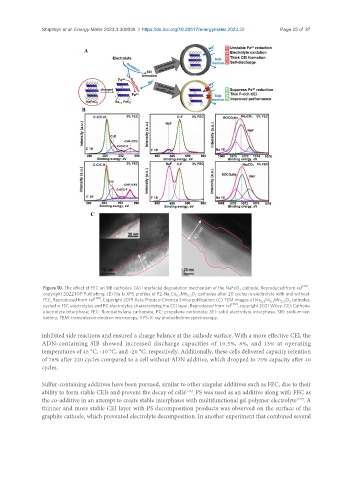
Figure 10. The effect of FEC on SIB cathodes. (A) Interfacial degradation mechanism of the NaFeO cathode, Reproduced from ref [20] ,
2
copyright 2022 IOP Publishing; (B) Na 1s XPS profiles of P2-Na Co Mn O cathodes after 20 cycles in electrolyte with and without
x 0.7 0.3 2
FEC, Reproduced from ref [129] , Copyright 2019 Acta Physico-Chimica Sinica publication; (C) TEM images of Na Ni Mn O cathodes
2/3 1/3 2/3 2
cycled in FEC electrolytes and PC electrolytes characterizing the CEI layer, Reproduced from ref [130] , copyright 2021 Wiley. CEI: Cathode
electrolyte interphase; FEC: fluoroethylene carbonate; PC: propylene carbonate; SEI: solid electrolyte interphase; SIB: sodium-ion
battery; TEM: transmission electron microscopy; XPS: X-ray photoelectron spectroscopy.
inhibited side reactions and ensured a charge balance at the cathode surface. With a more effective CEI, the
ADN-containing SIB showed increased discharge capacities of 10.5%, 8%, and 13% at operating
temperatures of 45 °C, -10 °C, and -20 °C, respectively. Additionally, these cells delivered capacity retention
of 78% after 220 cycles compared to a cell without ADN additive, which dropped to 75% capacity after 40
cycles.
Sulfur-containing additives have been pursued, similar to other singular additives such as FEC, due to their
[136]
ability to form stable CEIs and prevent the decay of cells . PS was used as an additive along with FEC as
the co-additive in an attempt to create stable interphases with multifunctional gel polymer electrolyte . A
[137]
thinner and more stable CEI layer with PS decomposition products was observed on the surface of the
graphite cathode, which prevented electrolyte decomposition. In another experiment that combined several