Page 168 - Read Online
P. 168
Page 30 of 37 Shipitsyn et al. Energy Mater 2023;3:300038 https://dx.doi.org/10.20517/energymater.2023.22
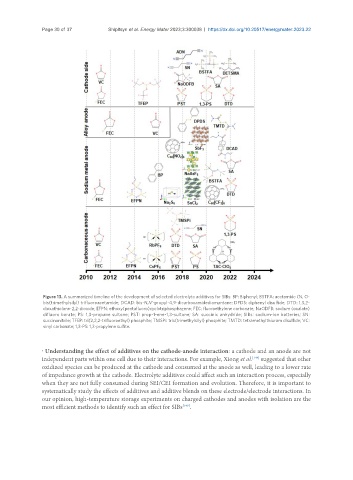
Figure 13. A summarized timeline of the development of selected electrolyte additives for SIBs. BP: Biphenyl; BSTFA: acetamide (N, O-
bis(trimethylsilyl) trifluoroacetamide; DCAD: bis-N,N’-propyl-4,9-dicarboxamidediamantane; DPDS: diphenyl disulfide; DTD: 1,3,2-
dioxathiolane-2,2-dioxide; EFPN: ethoxy(pentafluoro)cyclotriphosphazene; FEC: fluoroethylene carbonate; NaODFB: sodium (oxalate)
difluoro borate; PS: 1,3-propane sultone; PST: prop-1-ene-1,3-sultone; SA: succinic anhydride; SIBs: sodium-ion batteries; SN:
succinonitrile; TFEP: tri(2,2,2-trifluoroethyl) phosphite; TMSPi: tris(trimethylsilyl) phosphite; TMTD: tetramethylthiuram disulfide; VC:
vinyl carbonate; 1,3-PS: 1,3-propylene sulfite.
· Understanding the effect of additives on the cathode-anode interaction: a cathode and an anode are not
independent parts within one cell due to their interactions. For example, Xiong et al. suggested that other
[149]
oxidized species can be produced at the cathode and consumed at the anode as well, leading to a lower rate
of impedance growth at the cathode. Electrolyte additives could affect such an interaction process, especially
when they are not fully consumed during SEI/CEI formation and evolution. Therefore, it is important to
systematically study the effects of additives and additive blends on these electrode/electrode interactions. In
our opinion, high-temperature storage experiments on charged cathodes and anodes with isolation are the
most efficient methods to identify such an effect for SIBs .
[149]