Page 164 - Read Online
P. 164
Page 26 of 37 Shipitsyn et al. Energy Mater 2023;3:300038 https://dx.doi.org/10.20517/energymater.2023.22
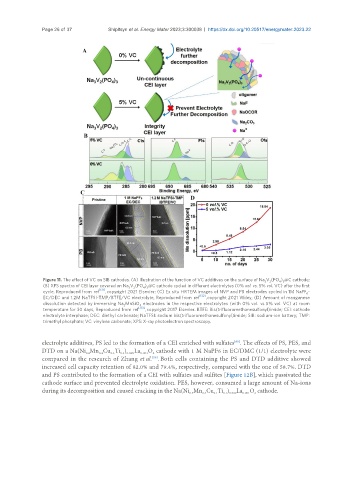
Figure 11. The effect of VC on SIB cathodes. (A) Illustration of the function of VC additives on the surface of Na V (PO ) @C cathode;
3 2 4 3
(B) XPS spectra of CEI layer covered on Na V (PO ) @C cathode cycled in different electrolytes (0% vol. vs. 5% vol. VC) after the first
3 2 4 3
[131]
cycle, Reproduced from ref , copyright 2021 Elsevier; (C) Ex situ HRTEM images of NVP and PB electrodes cycled in 1M NaPF -
6
[132]
EC/DEC and 1.2M NaTFSI-TMP/BTFE/VC electrolyte, Reproduced from ref , copyright 2021 Wiley; (D) Amount of manganese
dissolution detected by immersing Na MnSiO electrodes in the respective electrolytes (with 0% vol. vs. 5% vol. VC) at room
2 4
temperature for 30 days, Reproduced from ref [133] , copyright 2017 Elsevier. BTFE: Bis(trifluoromethanesulfonyl)imide; CEI: cathode
electrolyte interphase; DEC: diethyl carbonate; NaTFSI: sodium bis(trifluoromethanesulfonyl)imide; SIB: sodium-ion battery; TMP:
trimethyl phosphate; VC: vinylene carbonate; XPS: X-ray photoelectron spectroscopy.
electrolyte additives, PS led to the formation of a CEI enriched with sulfates . The effects of PS, PES, and
[69]
DTD on a Na(Ni Mn Cu Ti ) La 0.001 O cathode with 1 M NaPF6 in EC/DMC (1/1) electrolyte were
0.1 0.999
2
0.4
0.4
0.1
compared in the research of Zhang et al. . Both cells containing the PS and DTD additive showed
[59]
increased cell capacity retention of 82.0% and 79.4%, respectively, compared with the one of 58.7%. DTD
and PS contributed to the formation of a CEI with sulfates and sulfites [Figure 12B], which passivated the
cathode surface and prevented electrolyte oxidation. PES, however, consumed a large amount of Na-ions
during its decomposition and caused cracking in the Na(Ni Mn Cu Ti ) La O cathode.
0.001
2
0.4
0.4
0.1 0.999
0.1