Page 159 - Read Online
P. 159
Shipitsyn et al. Energy Mater 2023;3:300038 https://dx.doi.org/10.20517/energymater.2023.22 Page 21 of 37
Table 12. Other unsaturated chemical compounds as additives
Chemical name of the additive Fluoroethylene carbonate (FEC) Succinic anhydride (SA) Biphenyl (BP)
Chemical structure of additive
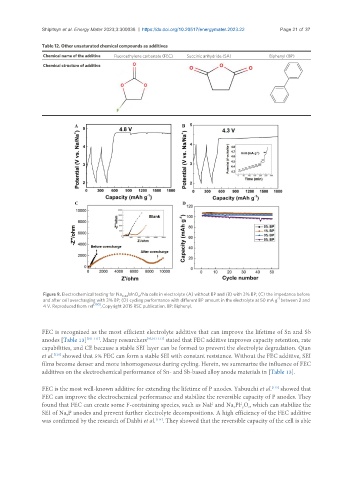
Figure 9. Electrochemical testing for Na 0.44 MnO /Na cells in electrolyte (A) without BP and (B) with 3% BP; (C) the impedance before
2
-1
and after cell overcharging with 3% BP; (D) cycling performance with different BP amount in the electrolyte at 50 mA g between 2 and
4 V. Reproduced from ref [100] , Copyright 2015 RSC publication. BP: Biphenyl.
FEC is recognized as the most efficient electrolyte additive that can improve the lifetime of Sn and Sb
anodes [Table 13] [101-111] . Many researchers [95,101-111] stated that FEC additive improves capacity retention, rate
capabilities, and CE because a stable SEI layer can be formed to prevent the electrolyte degradation. Qian
[110]
et al. showed that 5% FEC can form a stable SEI with constant resistance. Without the FEC additive, SEI
films become denser and more inhomogeneous during cycling. Herein, we summarize the influence of FEC
additives on the electrochemical performance of Sn- and Sb-based alloy anode materials in [Table 13].
[113]
FEC is the most well-known additive for extending the lifetime of P anodes. Yabuuchi et al. showed that
FEC can improve the electrochemical performance and stabilize the reversible capacity of P anodes. They
found that FEC can create some F-containing species, such as NaF and Na PF O , which can stabilize the
x
z
y
SEI of Na P anodes and prevent further electrolyte decompositions. A high efficiency of the FEC additive
x
was confirmed by the research of Dahbi et al. . They showed that the reversible capacity of the cell is able
[114]