Page 160 - Read Online
P. 160
Page 22 of 37 Shipitsyn et al. Energy Mater 2023;3:300038 https://dx.doi.org/10.20517/energymater.2023.22
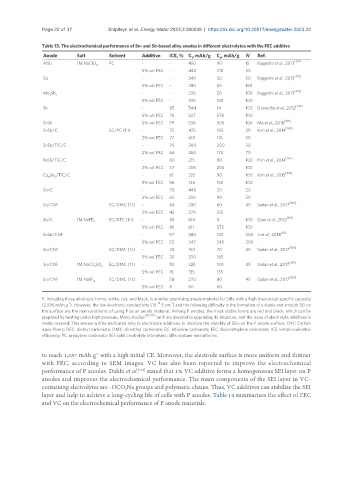
Table 13. The electrochemical performance of Sn- and Sb-based alloy anodes in different electrolytes with the FEC additive
Anode Salt Solvent Additive ICE, % C , mAh/g C , mAh/g N Ref.
1
n
AlSb 1M NaClO 4 PC - - 450 90 15 Baggetto et al., 2013 [101]
5% vol. FEC - 440 210 50
[102]
Ge - - 340 30 50 Baggetto et al., 2013
5% vol. FEC - 280 50 150
[103]
Mo Sb 7 - - 330 20 100 Baggetto et al., 2013
3
5% vol. FEC - 330 150 100
[104]
Sb - 65 544 14 100 Darwiche et al., 2012
5% vol. FEC 76 537 576 100
[105]
SnSb 5% vol. FEC 59 536 506 100 Ma et al., 2018
SnSb/C EC/PC (1:1) - 75 475 105 30 Kim et al., 2014 [106]
2% vol. FEC 77 610 110 30
SnSb/TiC/C - 70 300 200 30
2% vol. FEC 66 260 170 70
[107]
FeSb/TiC/C - 60 215 90 100 Kim et al., 2014
2% vol. FEC 57 205 204 100
[108]
Cu Sn /TiC/C - 61 225 90 100 Kim et al., 2015
6
5
2% vol. FEC 56 146 155 100
Sn/C - 70 445 20 20
2% vol. FEC 65 250 90 50
[109]
Sn/CNF EC/DMC (1:1) - 68 280 60 45 Sadan et al., 2017
5% vol. FEC 45 270 155
[110]
Sb/C 1M NaPF EC/DEC (1:1) - 85 610 0 100 Qian et al., 2012
6
5% vol. FEC 85 611 575 100
[111]
SnSb/CNF - 57 380 130 200 Ji et al., 2014
5% vol. FEC 53 347 345 200
[109]
Sn/CNF EC/DMC (1:1) - 24 150 70 45 Sadan et al., 2017
5% vol. FEC 30 230 185
[109]
Sn/CNF 1M NaCF SO EC/DMC (1:1) - 50 325 100 45 Sadan et al., 2017
3 3
5% vol. FEC 16 135 135
Sn/CNF 1M NaBF 4 EC/DMC (1:1) - 58 270 80 45 Sadan et al., 2017 [109]
5% vol. FEC 8 60 60
P, including three allotropic forms: white, red, and black, is another promising anode material for SIBs with a high theoretical specific capacity
-14
-1
-1
(2,596 mAh g ). However, the low electronic conductivity (10 S cm ) and the following difficulty in the formation of a stable and smooth SEI on
the surface are the main problems of using P as an anode material. Among P anodes, the most stable forms are red and black, which can be
[112-117]
prepared by heating under high pressure. Many studies on P are devoted to upgrading its structure, and the issue of electrolyte additives is
rarely covered. This review will be dedicated only to electrolyte additives to improve the stability of SEIs on the P anode surface. CNF: Carbon
nano-fibers; DEC: diethyl carbonate; DMC: dimethyl carbonate; EC: ethylene carbonate; FEC: fluoroethylene carbonate; ICE: initial coulombic
efficiency; PC: propylene carbonate; SEI: solid electrolyte interphase; SIBs: sodium-ion batteries.
to reach 1,587 mAh g with a high initial CE. Moreover, the electrode surface is more uniform and thinner
-1
with FEC, according to SEM images. VC has also been reported to improve the electrochemical
[114]
performance of P anodes. Dahbi et al. stated that 1% VC additive forms a homogeneous SEI layer on P
anodes and improves the electrochemical performance. The main components of the SEI layer in VC-
containing electrolytes are -OCO Na groups and polymeric chains. Thus, VC additives can stabilize the SEI
2
layer and help to achieve a long-cycling life of cells with P anodes. Table 14 summarizes the effect of FEC
and VC on the electrochemical performance of P anode materials.