Page 154 - Read Online
P. 154
Page 16 of 37 Shipitsyn et al. Energy Mater 2023;3:300038 https://dx.doi.org/10.20517/energymater.2023.22
Table 7. Ionic additives
Chemical name of the additive Trisaminocyclopropenium perchlorate (TAC·ClO ) RbPF 6 CsPF 6
4
Chemical structure of additive
Table 8. Ionic additives
Chemical name Tin Chloride (SnCl ) Sodium Polysulfide (Na S ) Antimony trifluoride (SbF ) Sodium Hexafluoroarsenate
2
2 6
3
of the additive (NaAsF )
6
Chemical
structure of
additive
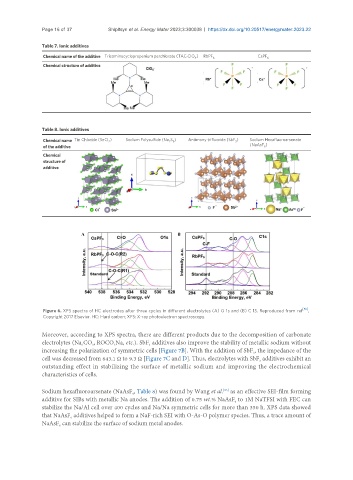
Figure 6. XPS spectra of HC electrodes after three cycles in different electrolytes (A) O 1s and (B) C 1S. Reproduced from ref [65] ,
Copyright 2017 Elsevier. HC: Hard carbon; XPS: X-ray photoelectron spectroscopy.
Moreover, according to XPS spectra, there are different products due to the decomposition of carbonate
electrolytes (Na CO , ROCO Na, etc.). SbF additives also improve the stability of metallic sodium without
2
3
2
3
increasing the polarization of symmetric cells [Figure 7B]. With the addition of SbF , the impedance of the
3
cell was decreased from 643.1 Ω to 9.3 Ω [Figure 7C and D]. Thus, electrolytes with SbF additives exhibit an
3
outstanding effect in stabilizing the surface of metallic sodium and improving the electrochemical
characteristics of cells.
Sodium hexafluoroarsenate (NaAsF , Table 8) was found by Wang et al. as an effective SEI-film forming
[85]
6
additive for SIBs with metallic Na anodes. The addition of 0.75 wt.% NaAsF to 1M NaTFSI with FEC can
6
stabilize the Na/Al cell over 400 cycles and Na/Na symmetric cells for more than 350 h. XPS data showed
that NaAsF additives helped to form a NaF-rich SEI with O-As-O polymer species. Thus, a trace amount of
6
NaAsF can stabilize the surface of sodium metal anodes.
6