Page 151 - Read Online
P. 151
Shipitsyn et al. Energy Mater 2023;3:300038 https://dx.doi.org/10.20517/energymater.2023.22 Page 13 of 37
Table 5. Phosphorus-containing additives
Chemical name of the additive Ethoxy(pentafluoro)cyclotriphosphazene (EFPN) Tris(trimethylsilyl) phosphite (TMSPi)
Chemical structure of additive
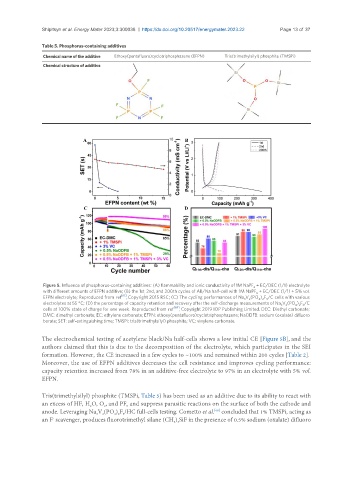
Figure 5. Influence of phosphorus-containing additives: (A) flammability and ionic conductivity of 1M NaPF + EC/DEC (1/1) electrolyte
6
with different amounts of EFPN additive; (B) the 1st 2nd, and 200th cycles of AB/Na half-cell with 1M NaPF + EC/DEC (1/1) + 5% vol.
,
6
EFPN electrolyte; Reproduced from ref [61] , Copyright 2015 RSC; (C) The cycling performances of Na V (PO ) F /C cells with various
2
4 2 3
3
electrolytes at 55 °C; (D) the percentage of capacity retention and recovery after the self-discharge measurements of Na V (PO ) F /C
4 2 3
2
3
cells at 100% state of charge for one week. Reproduced from ref [68] , Copyright 2019 IOP Publishing Limited. DEC: Diethyl carbonate;
DMC: dimethyl carbonate; EC: ethylene carbonate; EFPN: ethoxy(pentafluoro)cyclotriphosphazene; NaODFB: sodium (oxalate) difluoro
borate; SET: self-extinguishing time; TMSPi: tris(trimethylsilyl) phosphite; VC: vinylene carbonate.
The electrochemical testing of acetylene black/Na half-cells shows a low initial CE [Figure 5B], and the
authors claimed that this is due to the decomposition of the electrolyte, which participates in the SEI
formation. However, the CE increased in a few cycles to ~100% and remained within 200 cycles [Table 2].
Moreover, the use of EFPN additives decreases the cell resistance and improves cycling performance:
capacity retention increased from 78% in an additive-free electrolyte to 97% in an electrolyte with 5% vol.
EFPN.
Tris(trimethylsilyl) phosphite (TMSPi, Table 5) has been used as an additive due to its ability to react with
an excess of HF, H O, O , and PF and suppress parasitic reactions on the surface of both the cathode and
2
2
5
anode. Leveraging Na V (PO ) F /HC full-cells testing. Cometto et al. concluded that 1% TMSPi, acting as
[68]
2
3
4 2 3
-
an F scavenger, produces fluorotrimethyl silane (CH ) SiF in the presence of 0.5% sodium (oxalate) difluoro
3 3