Page 148 - Read Online
P. 148
Page 10 of 37 Shipitsyn et al. Energy Mater 2023;3:300038 https://dx.doi.org/10.20517/energymater.2023.22
Table 3. Fluorine-containing additives
Chemical name of the additive Fluoroethylene carbonate (FEC) Difluoroethylene carbonate (DFEC)
Chemical structure of additive
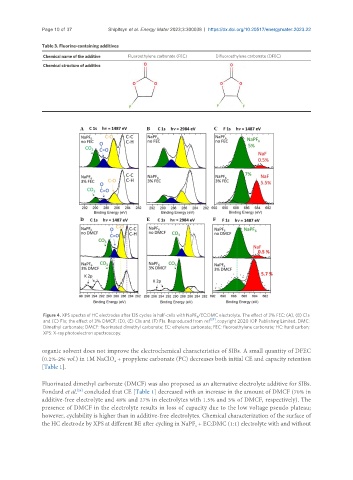
Figure 4. XPS spectra of HC electrodes after 135 cycles in half-cells with NaPF /EC:DMC electrolyte. The effect of 3% FEC: (A), (B) C1s
6
and (C) F1s; the effect of 3% DMCF: (D), (E) C1s and (F) F1s. Reproduced from ref [24] , copyright 2020 IOP Publishing Limited. DMC:
Dimethyl carbonate; DMCF: fluorinated dimethyl carbonate; EC: ethylene carbonate; FEC: fluoroethylene carbonate; HC: hard carbon;
XPS: X-ray photoelectron spectroscopy.
organic solvent does not improve the electrochemical characteristics of SIBs. A small quantity of DFEC
(0.2%-2% vol.) in 1M NaClO + propylene carbonate (PC) decreases both initial CE and capacity retention
4
[Table 1].
Fluorinated dimethyl carbonate (DMCF) was also proposed as an alternative electrolyte additive for SIBs.
Fondard et al. concluded that CE [Table 1] decreased with an increase in the amount of DMCF (70% in
[24]
additive-free electrolyte and 48% and 27% in electrolytes with 1.5% and 3% of DMCF, respectively). The
presence of DMCF in the electrolyte results in loss of capacity due to the low voltage pseudo plateau;
however, cyclability is higher than in additive-free electrolytes. Chemical characterization of the surface of
the HC electrode by XPS at different BE after cycling in NaPF + EC:DMC (1:1) electrolyte with and without
6