Page 147 - Read Online
P. 147
Shipitsyn et al. Energy Mater 2023;3:300038 https://dx.doi.org/10.20517/energymater.2023.22 Page 9 of 37
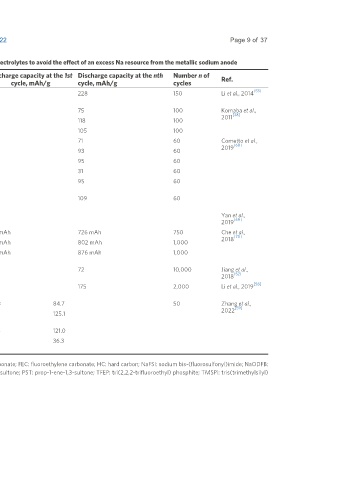
Table 2. A summary of the electrochemical performance of full cells with HC anodes in different electrolytes to avoid the effect of an excess Na resource from the metallic sodium anode
CE at the 1st Discharge capacity at the 1st Discharge capacity at the nth Number n of
Cathode Electrolyte Additive Ref.
cycle, % cycle, mAh/g cycle, mAh/g cycles
[53]
Na 2/3 Ni Mn 2/3 O 2 1M NaClO + EC/DEC - 76 300 228 150 Li et al., 2014
1/3
4
(1:1)
NaNi Mn O 1M NaClO + PC - - 215 75 100 Komaba et al.,
1/2 1/2 2 4 [38]
2011
1M NaPF + PC - - 247 118 100
6
1M NaTFSA + PC - - 249 105 100
Na V (PO ) F 1M NaPF + EC/DMC - 83.0 109 71 60 Cometto et al.,
3 2 4 2 3 6 [68]
(1:1) 2019
1% TMSPi 76.0 110 93 60
3% VC 88.0 108 95 60
0.5% NaODFB 85.0 107 31 60
0.5% NaODFB + 1% 70.0 109 95 60
TMSPi
0.5% NaODFB + 1% 83.0 110 109 60
TMSPi + 3% VC
Na V (PO ) F 1M NaPF + EC/PC (1:1) - Yan et al.,
6
3
4 2 3
2
2019 [69]
NaNi Fe Mn O 1M NaPF + PC/EMC 2% FEC - 950 mAh 726 mAh 750 Che et al.,
1/3 1/3 1/3 2 6 [70]
(1:1) 2018
2% FEC + 1% PST - 950 mAh 802 mAh 1,000
2% FEC + 1% PST + 1% - 950 mAh 876 mAh 1,000
DTD
Na V (PO ) 0.9M NaFSI + TFEP - 70.6 87 72 10,000 Jiang et al.,
3 2 4 3 [52]
2018
[58]
Na[Cu Ni 2/9 Fe Mn 1/3 1M NaClO + 2% FEC 74.0 252 175 2,000 Li et al., 2019
1/9
1/3
4
]O EC/DMC/PC (1:1:1)
2
Na(Ni Mn Cu Ti ) 1M NaPF + EC/DMC 2% wt. FEC 72.3 144.3 84.7 50 Zhang et al.,
0.4 0.4 0.1 0.1 6 [59]
La O (1:1) 2022
0.999 0.001 2 2% wt. FEC + 2% wt. 1,3- 76.7 152.6 125.1
PS
2% wt. FEC + 2% wt. DTD 79.1 152.4 121.0
2% wt. FEC + 0.2% wt. 65.3 139.2 36.3
PES
DEC: Diethyl carbonate; DMC: dimethyl carbonate; DTD: 1,3,2-dioxathiolane-2,2-dioxide; EC: ethylene carbonate; FEC: fluoroethylene carbonate; HC: hard carbon; NaFSI: sodium bis-(fluorosulfonyl)imide; NaODFB:
sodium (oxalate) difluoro borate; PC: propylene carbonate; PES: prop-1-ene-1,3-sultone; PS: 1,3-propane sultone; PST: prop-1-ene-1,3-sultone; TFEP: tri(2,2,2-trifluoroethyl) phosphite; TMSPi: tris(trimethylsilyl)
phosphite; VC: vinyl carbonate; 1,3-PS: 1,3-propylene sulfite.