Page 47 - Read Online
P. 47
Aydin et al. J Transl Genet Genom. 2025;9:406-26 https://dx.doi.org/10.20517/jtgg.2025.108 Page 414
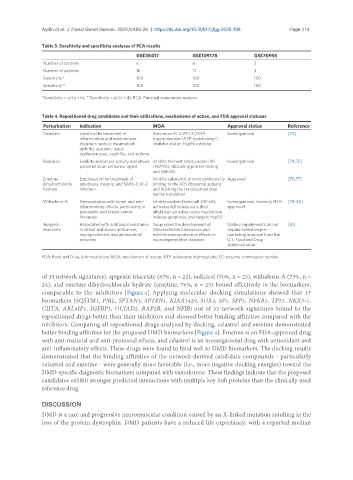
Table 3. Sensitivity and specificity analyses of PCA results
GSE38417 GSE109178 GSE70955
Number of controls 6 6 3
Number of patients 16 17 3
Sensitivity* 100 100 100
Specificity** 100 100 100
*Sensitivity = a/(a + b), **Specificity = d/(c + d). PCA: Principal component analysis.
Table 4. Repositioned drug candidates and their utilizations, mechanisms of action, and FDA approval statuses
Perturbation Indication MOA Approval status Reference
Celastrol Used for the treatment of Acts as an EC 5.99.1.3 [DNA Investigational [73]
inflammation and autoimmune topoisomerase (ATP-hydrolysing)]
disorders such as rheumatoid inhibitor and an Hsp90 inhibitor
arthritis, systemic lupus
erythematosus, nephritis, and asthma
Radicicol Exhibits anticancer activity and shows Inhibits the heat shock protein 90 Investigational [74,75]
potential as an antitumor agent (HSP90), disrupting protein folding
and stability
Emetine Employed in the treatment of Inhibits eukaryotic protein synthesis by Approved [76,77]
dihydrochloride amebiasis, malaria, and SARS-CoV-2 binding to the 40S ribosomal subunit
hydrate infection and blocking the translocation step
during translation
Withaferin-A Demonstrates anti-tumor and anti- Inhibits nuclear factor-κB (NF-κB), Investigational, formerly FDA- [78-80]
inflammatory effects, particularly in activates IκB kinase via a thiol approved
pancreatic and breast cancer alkylation-sensitive redox mechanism,
therapies induces apoptosis, and targets Hsp90
Apigenin Associated with antifungal resistance Suppresses the development of Dietary supplements do not [81]
triacetate in wheat and shows anticancer, Mycobacterium tuberculosis and require extensive pre-
neuroprotective, and antibacterial exhibits neuroprotective effects in marketing approval from the
activities neurodegenerative diseases U.S. Food and Drug
Administration
FDA: Food and Drug Administration; MOA: mechanism of action; ATP: adenosine triphosphate; EC: enzyme commission number.
of 33 network signatures), apigenin triacetate (67%, n = 22), radicicol (70%, n = 23), withaferin-A (73%, n =
24), and emetine dihydrochloride hydrate (emetine, 76%, n = 25) bound effectively to the biomarkers,
comparable to the inhibitors [Figure 4]. Applying molecular docking simulations showed that 17
biomarkers (SQSTM1, PML, SPTAN1, SPTBN1, KIAA1429, SOX4, SP1, SPP1, NFKB1, TP53, NKX3-1,
CIITA, ARL6IP1, IGFBP5, OCIAD2, RAP2B, and NFIB) out of 33 network signatures bound to the
repositioned drugs better than their inhibitors and showed better binding affinities compared with the
inhibitors. Comparing all repositioned drugs analyzed by docking, celastrol and emetine demonstrated
better binding affinities for the proposed DMD biomarkers [Figure 4]. Emetine is an FDA-approved drug
with anti-malarial and anti-protozoal effects, and celastrol is an investigational drug with antioxidant and
anti-inflammatory effects. These drugs were found to bind well to DMD biomarkers. The docking results
demonstrated that the binding affinities of the network-derived candidate compounds - particularly
celastrol and emetine - were generally more favorable (i.e., more negative docking energies) toward the
DMD-specific diagnostic biomarkers compared with vamolorone. These findings indicate that the proposed
candidates exhibit stronger predicted interactions with multiple key hub proteins than the clinically used
reference drug.
DISCUSSION
DMD is a rare and progressive neuromuscular condition caused by an X-linked mutation resulting in the
loss of the protein dystrophin. DMD patients have a reduced life expectancy, with a reported median