Page 183 - Read Online
P. 183
Page 8 of 14 Wang et al. Energy Mater 2023;3:300040 https://dx.doi.org/10.20517/energymater.2023.28
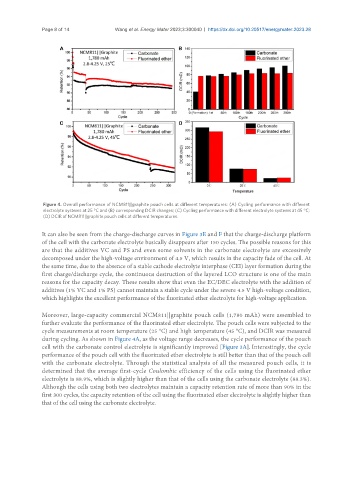
Figure 4. Overall performance of NCM811||graphite pouch cells at different temperatures: (A) Cycling performance with different
electrolyte systems at 25 °C and (B) corresponding DCIR changes; (C) Cycling performance with different electrolyte systems at 45 °C;
(D) DCIR of NCM811||graphite pouch cells at different temperatures.
It can also be seen from the charge-discharge curves in Figure 3E and F that the charge-discharge platform
of the cell with the carbonate electrolyte basically disappears after 150 cycles. The possible reasons for this
are that the additives VC and PS and even some solvents in the carbonate electrolyte are excessively
decomposed under the high-voltage environment of 4.5 V, which results in the capacity fade of the cell. At
the same time, due to the absence of a stable cathode electrolyte interphase (CEI) layer formation during the
first charge/discharge cycle, the continuous destruction of the layered LCO structure is one of the main
reasons for the capacity decay. These results show that even the EC/DEC electrolyte with the addition of
additives (1% VC and 1% PS) cannot maintain a stable cycle under the severe 4.5 V high-voltage condition,
which highlights the excellent performance of the fluorinated ether electrolyte for high-voltage application.
Moreover, large-capacity commercial NCM811||graphite pouch cells (1,780 mAh) were assembled to
further evaluate the performance of the fluorinated ether electrolyte. The pouch cells were subjected to the
cycle measurements at room temperature (25 °C) and high temperature (45 °C), and DCIR was measured
during cycling. As shown in Figure 4A, as the voltage range decreases, the cycle performance of the pouch
cell with the carbonate control electrolyte is significantly improved [Figure 3A]. Interestingly, the cycle
performance of the pouch cell with the fluorinated ether electrolyte is still better than that of the pouch cell
with the carbonate electrolyte. Through the statistical analysis of all the measured pouch cells, it is
determined that the average first-cycle Coulombic efficiency of the cells using the fluorinated ether
electrolyte is 88.9%, which is slightly higher than that of the cells using the carbonate electrolyte (88.3%).
Although the cells using both two electrolytes maintain a capacity retention rate of more than 90% in the
first 300 cycles, the capacity retention of the cell using the fluorinated ether electrolyte is slightly higher than
that of the cell using the carbonate electrolyte.