Page 180 - Read Online
P. 180
Wang et al. Energy Mater 2023;3:300040 https://dx.doi.org/10.20517/energymater.2023.28 Page 5 of 14
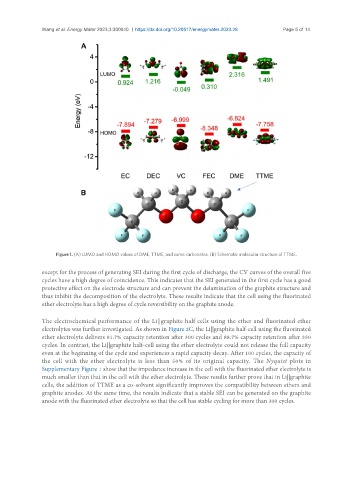
Figure 1. (A) LUMO and HOMO values of DME, TTME, and some carbonates. (B) Schematic molecular structure of TTME.
except for the process of generating SEI during the first cycle of discharge, the CV curves of the overall five
cycles have a high degree of coincidence. This indicates that the SEI generated in the first cycle has a good
protective effect on the electrode structure and can prevent the delamination of the graphite structure and
thus inhibit the decomposition of the electrolyte. These results indicate that the cell using the fluorinated
ether electrolyte has a high degree of cycle reversibility on the graphite anode.
The electrochemical performance of the Li||graphite half cells using the ether and fluorinated ether
electrolytes was further investigated. As shown in Figure 2C, the Li||graphite half-cell using the fluorinated
ether electrolyte delivers 91.7% capacity retention after 300 cycles and 88.7% capacity retention after 350
cycles. In contrast, the Li||graphite half-cell using the ether electrolyte could not release the full capacity
even at the beginning of the cycle and experiences a rapid capacity decay. After 100 cycles, the capacity of
the cell with the ether electrolyte is less than 50% of its original capacity. The Nyquist plots in
Supplementary Figure 1 show that the impedance increase in the cell with the fluorinated ether electrolyte is
much smaller than that in the cell with the ether electrolyte. These results further prove that in Li||graphite
cells, the addition of TTME as a co-solvent significantly improves the compatibility between ethers and
graphite anodes. At the same time, the results indicate that a stable SEI can be generated on the graphite
anode with the fluorinated ether electrolyte so that the cell has stable cycling for more than 300 cycles.