Page 182 - Read Online
P. 182
Wang et al. Energy Mater 2023;3:300040 https://dx.doi.org/10.20517/energymater.2023.28 Page 7 of 14
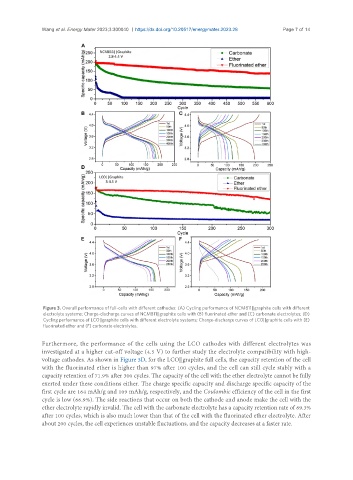
Figure 3. Overall performance of full-cells with different cathodes: (A) Cycling performance of NCM811||graphite cells with different
electrolyte systems; Charge-discharge curves of NCM811||graphite cells with (B) fluorinated ether and (C) carbonate electrolytes; (D)
Cycling performance of LCO||graphite cells with different electrolyte systems; Charge-discharge curves of LCO||graphite cells with (E)
fluorinated ether and (F) carbonate electrolytes.
Furthermore, the performance of the cells using the LCO cathodes with different electrolytes was
investigated at a higher cut-off voltage (4.5 V) to further study the electrolyte compatibility with high-
voltage cathodes. As shown in Figure 3D, for the LCO||graphite full cells, the capacity retention of the cell
with the fluorinated ether is higher than 97% after 100 cycles, and the cell can still cycle stably with a
capacity retention of 71.9% after 300 cycles. The capacity of the cell with the ether electrolyte cannot be fully
exerted under these conditions either. The charge specific capacity and discharge specific capacity of the
first cycle are 164 mAh/g and 109 mAh/g, respectively, and the Coulombic efficiency of the cell in the first
cycle is low (66.9%). The side reactions that occur on both the cathode and anode make the cell with the
ether electrolyte rapidly invalid. The cell with the carbonate electrolyte has a capacity retention rate of 69.3%
after 100 cycles, which is also much lower than that of the cell with the fluorinated ether electrolyte. After
about 200 cycles, the cell experiences unstable fluctuations, and the capacity decreases at a faster rate.