Page 186 - Read Online
P. 186
Wang et al. Energy Mater 2023;3:300040 https://dx.doi.org/10.20517/energymater.2023.28 Page 11 of 14
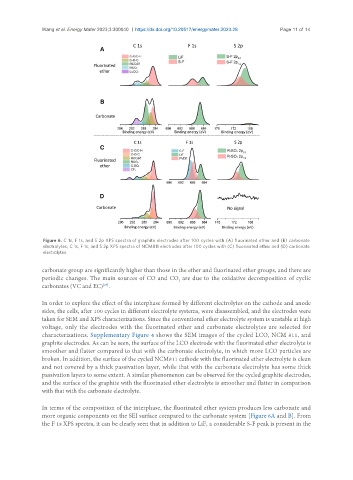
Figure 6. C 1s, F 1s, and S 2p XPS spectra of graphite electrodes after 100 cycles with (A) fluorinated ether and (B) carbonate
electrolytes; C 1s, F 1s, and S 2p XPS spectra of NCM811 electrodes after 100 cycles with (C) fluorinated ether and (D) carbonate
electrolytes.
carbonate group are significantly higher than those in the ether and fluorinated ether groups, and there are
periodic changes. The main sources of CO and CO are due to the oxidative decomposition of cyclic
2
carbonates (VC and EC) .
[27]
In order to explore the effect of the interphase formed by different electrolytes on the cathode and anode
sides, the cells, after 100 cycles in different electrolyte systems, were disassembled, and the electrodes were
taken for SEM and XPS characterizations. Since the conventional ether electrolyte system is unstable at high
voltage, only the electrodes with the fluorinated ether and carbonate electrolytes are selected for
characterizations. Supplementary Figure 4 shows the SEM images of the cycled LCO, NCM 811, and
graphite electrodes. As can be seen, the surface of the LCO electrode with the fluorinated ether electrolyte is
smoother and flatter compared to that with the carbonate electrolyte, in which more LCO particles are
broken. In addition, the surface of the cycled NCM811 cathode with the fluorinated ether electrolyte is clean
and not covered by a thick passivation layer, while that with the carbonate electrolyte has some thick
passivation layers to some extent. A similar phenomenon can be observed for the cycled graphite electrodes,
and the surface of the graphite with the fluorinated ether electrolyte is smoother and flatter in comparison
with that with the carbonate electrolyte.
In terms of the composition of the interphase, the fluorinated ether system produces less carbonate and
more organic components on the SEI surface compared to the carbonate system [Figure 6A and B]. From
the F 1s XPS spectra, it can be clearly seen that in addition to LiF, a considerable S-F peak is present in the