Page 181 - Read Online
P. 181
Page 6 of 14 Wang et al. Energy Mater 2023;3:300040 https://dx.doi.org/10.20517/energymater.2023.28
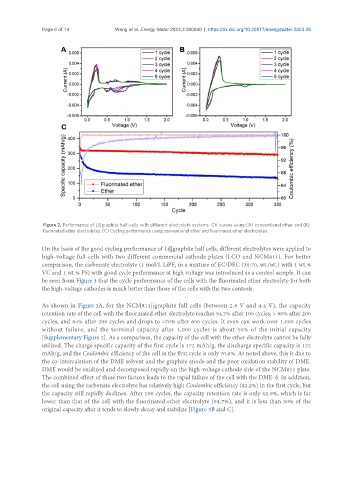
Figure 2. Performance of Li||graphite half-cells with different electrolyte systems: CV curves using (A) conventional ether and (B)
fluorinated ether electrolytes; (C) Cycling performance using conventional ether and fluorinated ether electrolytes.
On the basis of the good cycling performance of Li||graphite half cells, different electrolytes were applied to
high-voltage full-cells with two different commercial cathode plates (LCO and NCM811). For better
comparison, the carbonate electrolyte (1 mol/L LiPF in a mixture of EC/DEC (30:70, wt./wt.) with 1 wt.%
6
VC and 1 wt.% PS) with good cycle performance at high voltage was introduced as a control sample. It can
be seen from Figure 3 that the cycle performance of the cells with the fluorinated ether electrolyte for both
the high-voltage cathodes is much better than those of the cells with the two controls.
As shown in Figure 3A, for the NCM811||graphite full cells (between 2.8 V and 4.4 V), the capacity
retention rate of the cell with the fluorinated ether electrolyte reaches 94.7% after 100 cycles, > 90% after 200
cycles, and 84% after 300 cycles and drops to ≈70% after 600 cycles. It even can work over 1,000 cycles
without failure, and the terminal capacity after 1,000 cycles is about 50% of the initial capacity
[Supplementary Figure 2]. As a comparison, the capacity of the cell with the ether electrolyte cannot be fully
utilized. The charge specific capacity of the first cycle is 172 mAh/g, the discharge specific capacity is 122
mAh/g, and the Coulombic efficiency of the cell in the first cycle is only 70.8%. As noted above, this is due to
the co-intercalation of the DME solvent and the graphite anode and the poor oxidation stability of DME.
DME would be oxidized and decomposed rapidly on the high-voltage cathode side of the NCM811 plate.
The combined effect of these two factors leads to the rapid failure of the cell with the DME-d. In addition,
the cell using the carbonate electrolyte has relatively high Coulombic efficiency (82.2%) in the first cycle, but
the capacity still rapidly declines. After 100 cycles, the capacity retention rate is only 62.9%, which is far
lower than that of the cell with the fluorinated ether electrolyte (94.7%), and it is less than 30% of the
original capacity after it tends to slowly decay and stabilize [Figure 3B and C].