Page 354 - Read Online
P. 354
Kanevsky et al. Stretch device for scar therapy
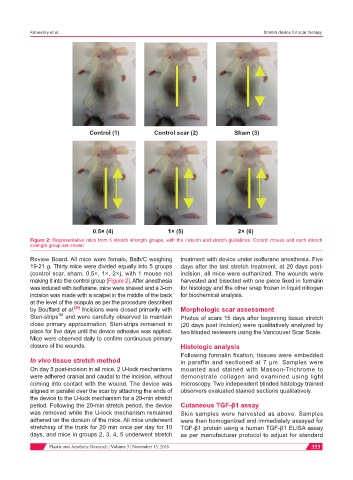
Control (1) Control scar (2) Sham (3)
0.5× (4) 1× (5) 2× (6)
Figure 2: Representative mice from 5 stretch strength groups, with the incision and stretch guidelines. Control mouse and each stretch
strength group are shown
Review Board. All mice were female, Balb/C weighing treatment with device under isoflurane anesthesia. Five
19-21 g. Thirty mice were divided equally into 5 groups days after the last stretch treatment, at 20 days post-
(control scar, sham, 0.5×, 1×, 2×), with 1 mouse not incision, all mice were euthanized. The wounds were
making it into the control group [Figure 2]. After anesthesia harvested and bisected with one piece fixed in formalin
was induced with isoflurane, mice were shaved and a 3-cm for histology and the other snap frozen in liquid nitrogen
incision was made with a scalpel in the middle of the back for biochemical analysis.
at the level of the scapula as per the procedure described
[29]
by Bouffard et al. Incisions were closed primarily with Morphologic scar assessment
TM
Steri-strips and were carefully observed to maintain Photos of scars 15 days after beginning tissue stretch
close primary approximation. Steri-strips remained in (20 days post incision) were qualitatively analyzed by
place for five days until the device adhesive was applied. two blinded reviewers using the Vancouver Scar Scale.
Mice were observed daily to confirm continuous primary
closure of the wounds. Histologic analysis
Following formalin fixation, tissues were embedded
In vivo tissue stretch method in paraffin and sectioned at 7 μm. Samples were
On day 5 post-incision in all mice, 2 U-lock mechanisms mounted and stained with Masson-Trichrome to
were adhered cranial and caudal to the incision, without demonstrate collagen and examined using light
coming into contact with the wound. The device was microscopy. Two independent blinded histology trained
aligned in parallel over the scar by attaching the ends of observers evaluated stained sections qualitatively.
the device to the U-lock mechanism for a 20-min stretch
period. Following the 20-min stretch period, the device Cutaneous TGF-β1 assay
was removed while the U-lock mechanism remained Skin samples were harvested as above. Samples
adhered on the dorsum of the mice. All mice underwent were then homogenized and immediately assayed for
stretching of the trunk for 20 min once per day for 10 TGF-β1 protein using a human TGF-β1 ELISA assay
days, and mice in groups 2, 3, 4, 5 underwent stretch as per manufacturer protocol to adjust for standard
Plastic and Aesthetic Research ¦ Volume 3 ¦ November 15, 2016 353