Page 47 - Read Online
P. 47
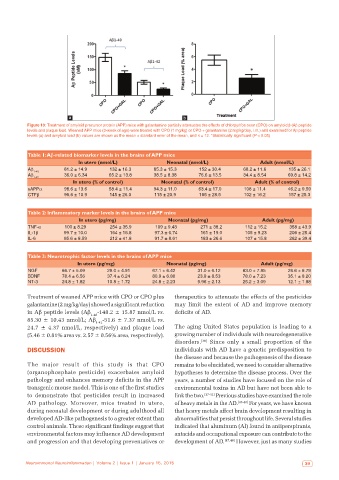
a b
Figure 10: Treatment of amyloid precursor protein (APP) mice with galantamine partially attenuates the effects of chlorpyrifos oxon (CPO) on amyloid β (Aβ peptide
levels and plaque load. Weaned APP mice (3‑week of age) were treated with CPO (1 mg/kg) or CPO + galantamine (2 mg/kg/day, i.m.) and examined for Aβ peptide
levels (a) and amyloid load (b) values are shown as the mean ± standard error of the mean, and n = 12. *Statistically significant (P < 0.05)
Table 1: Aβ‑related biomarker levels in the brains of APP mice
In utero (nmol/L) Neonatal (nmol/L) Adult (nmol/L)
Aβ 1‑40 81.2 ± 14.9 132 ± 16.3 85.3 ± 15.3 152 ± 30.4 68.2 ± 11.6 135 ± 26.1
Aβ 1‑42 36.0 ± 6.34 65.2 ± 13.8 38.5 ± 8.38 76.6 ± 13.5 34.4 ± 8.54 69.8 ± 14.2
In utero (% of control) Neonatal (% of control) Adult (% of control)
sAPPα 98.6 ± 13.6 58.4 ± 11.4 94.3 ± 11.0 63.4 ± 17.0 108 ± 11.4 46.2 ± 9.59
CTFβ 96.6 ± 10.9 145 ± 26.0 115 ± 20.9 165 ± 28.5 102 ± 16.2 157 ± 25.3
Table 2: Inflammatory marker levels in the brains of APP mice
In utero (pg/mg) Neonatal (pg/mg) Adult (pg/mg)
TNF‑α 100 ± 8.29 254 ± 35.9 109 ± 9.43 271 ± 38.2 112 ± 15.2 358 ± 43.9
IL‑1β 99.7 ± 10.0 164 ± 16.8 97.3 ± 6.74 161 ± 19.0 105 ± 9.23 206 ± 25.4
IL‑6 95.6 ± 8.59 212 ± 41.8 91.7 ± 8.61 183 ± 26.6 107 ± 15.8 262 ± 39.4
Table 3: Neurotrophic factor levels in the brains of APP mice
In utero (pg/mg) Neonatal (pg/mg) Adult (pg/mg)
NGF 66.7 ± 5.09 29.0 ± 4.91 67.1 ± 6.42 31.0 ± 6.12 63.0 ± 7.95 26.6 ± 6.79
BDNF 78.4 ± 6.56 37.4 ± 6.24 80.9 ± 6.80 23.9 ± 8.53 78.0 ± 7.23 35.1 ± 8.20
NT‑3 24.8 ± 1.82 10.8 ± 1.72 24.8 ± 2.23 9.96 ± 2.13 25.2 ± 3.09 12.1 ± 1.88
Treatment of weaned APP mice with CPO or CPO plus therapeutics to attenuate the effects of the pesticides
galantamine (2 mg/kg/day) showed a significant reduction may limit the extent of AD and improve memory
in Aβ peptide levels (Aβ 1-40 -148.2 ± 15.87 nmol/L vs. deficits of AD.
85.30 ± 10.43 nmol/L; Aβ -51.6 ± 7.37 nmol/L vs.
1-42
24.7 ± 4.37 nmol/L, respectively) and plaque load The aging United States population is leading to a
(5.46 ± 0.81% area vs. 2.57 ± 0.56% area, respectively). growing number of individuals with neurodegenerative
disorders. [36] Since only a small proportion of the
DISCUSSION individuals with AD have a genetic predisposition to
the disease and because the pathogenesis of the disease
The major result of this study is that CPO remains to be elucidated, we need to consider alternative
(organophosphate pesticide) exacerbates amyloid hypotheses to determine the disease process. Over the
pathology and enhances memory deficits in the APP years, a number of studies have focused on the role of
transgenic mouse model. This is one of the first studies environmental toxins in AD but have not been able to
to demonstrate that pesticides result in increased link the two. [37-42] Previous studies have examined the role
AD pathology. Moreover, mice treated in utero, of heavy metals in the AD. [43-46] For years, we have known
during neonatal development or during adulthood all that heavy metals affect brain development resulting in
developed AD-like pathogenesis to a greater extent than abnormalities that persist throughout life. Several studies
control animals. These significant findings suggest that indicated that aluminum (Al) found in antiperspirants,
environmental factors may influence AD development antacids and occupational exposure can contribute to the
and progression and that developing preventatives or development of AD. [47,48] However, just as many studies
Neuroimmunol Neuroinflammation | Volume 2 | Issue 1 | January 15, 2015 39