Page 13 - Read Online
P. 13
Wang et al. J Transl Genet Genom 2023;7:126-40 https://dx.doi.org/10.20517/jtgg.2023.07 Page 134
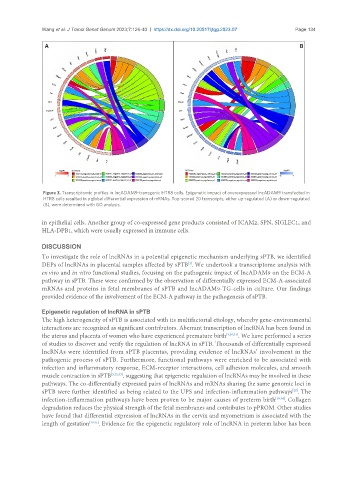
Figure 3. Transcriptomic profiles in IncADAM9-transgenic HTR8 cells. Epigenetic impact of overexpressed IncADAM9 transfected in
HTR8 cells resulted in a global differential expression of mRNAs. Top-scored 20 transcripts, either up-regulated (A) or down-regulated
(B), were determined with GO analysis.
in epithelial cells. Another group of co-expressed gene products consisted of ICAM2, SPN, SIGLEC1, and
HLA-DPB1, which were usually expressed in immune cells.
DISCUSSION
To investigate the role of lncRNAs in a potential epigenetic mechanism underlying sPTB, we identified
[3]
DEPs of lncRNAs in placental samples affected by sPTB . We undertook a transcriptome analysis with
ex vivo and in vitro functional studies, focusing on the pathogenic impact of lncADAM9 on the ECM-A
pathway in sPTB. These were confirmed by the observation of differentially expressed ECM-A-associated
mRNAs and proteins in fetal membranes of sPTB and lncADAM9-TG-cells in culture. Our findings
provided evidence of the involvement of the ECM-A pathway in the pathogenesis of sPTB.
Epigenetic regulation of lncRNA in sPTB
The high heterogeneity of sPTB is associated with its multifactorial etiology, whereby gene-environmental
interactions are recognized as significant contributors. Aberrant transcription of lncRNA has been found in
the uterus and placenta of women who have experienced premature birth [3,30,31] . We have performed a series
of studies to discover and verify the regulation of lncRNA in sPTB. Thousands of differentially expressed
lncRNAs were identified from sPTB placentas, providing evidence of lncRNAs’ involvement in the
pathogenic process of sPTB. Furthermore, functional pathways were enriched to be associated with
infection and inflammatory response, ECM-receptor interactions, cell adhesion molecules, and smooth
muscle contraction in sPTB [3,32,33] , suggesting that epigenetic regulation of lncRNAs may be involved in these
pathways. The co-differentially expressed pairs of lncRNAs and mRNAs sharing the same genomic loci in
sPTB were further identified as being related to the UPS and infection-inflammation pathways . The
[22]
infection-inflammation pathways have been proven to be major causes of preterm birth [18,34] . Collagen
degradation reduces the physical strength of the fetal membranes and contributes to pPROM. Other studies
have found that differential expression of lncRNAs in the cervix and myometrium is associated with the
length of gestation [30,31] . Evidence for the epigenetic regulatory role of lncRNA in preterm labor has been