Page 154 - Read Online
P. 154
Ieni et al. Colonic metastasis by a uterine LCNEC
in the uterine corpus involving the myometrium were present for CK, CK 7, CK20, CDX2, TTF1, Pax-
and serosal layer, with an infiltration of the colonic 8, CD10, vimentin, desmin and CD99. The growth
wall indicating lymph node metastases as well. The fraction, assessed with Ki67, revealed a positivity of
proliferation showed a uniform solid pattern, with more than 80% of neoplastic elements. A diagnosis
complete absence of glandular differentiation and of infiltrating poorly differentiated LCNEC was made,
areas of geographic necrosis [Figure 2A]; it was based on synaptophysin, chromogranin-A, ER and
characterized by medium and large-sized cells with PgR immunoreactivity. The diagnosis was classified as
hyperchromatic and pleomorphic nuclei, prominent a primary tumor of the uterus, with extensive colonic
nucleoli and high mitotic activity [Figure 2B]. and peritoneal spread. In light of these findings,
Immunohistochemistry revealed a diffuse positivity for we took the opportunity to re-examine the original
estrogen receptor (ER) [Figure 3A], chromogranin-A neoplastic paraffin-block taken at the colonic level
[Figure 3B], synaptophysin [Figure 3C], MLH1, MSH2, during the first surgical procedure. Histologically the
MSH6 and a partial staining for EMA, CD56 and colonic wall was extensively ab-extrinseco infiltrated
progesterone receptor (PgR). No immunostainings by a highly cellular solid proliferation [Figure 4A],
suggestive of a poorly differentiated adenocarcinoma,
but absolutely unreactive for CK20 [Figure 4B], a
marker usually positive in colonic cancer. Finally, a
heterogeneous, well evident, cytoplasmic staining for
chromogranin-A (Figure 4B, inset) was appreciable in
neoplastic elements. These morphological data were
consistent with a diagnosis of colonic parietal infiltration
by aggressive neuroendocrine carcinoma.
DISCUSSION
NETs are more generally identified in the
gastrointestinal tract, pancreas, lung and thymus,
while in the female reproductive tract they account
for about 2% of all gynecologic cancer. [6,7] According
to World Health Organization classification, NETs are
classified in two principal groups: poorly differentiated
neuroendocrine carcinomas (NECs) and well-
differentiated NETs. NECs include small and large
[8]
cell neuroendocrine carcinoma, while NETs include
typical and atypical carcinoids. [8]
Poorly differentiated LCNEC of the endometrium
is a very uncommon tumor representing only 0.8%
of endometrial cancers and they are considered
particularly aggressive neoplasms with a tendency
for early metastases and poor outcomes. Usually,
[9]
endometrial NECs are combined with other epithelial
neoplasms; in detail, 50-80% of cases are admixed with
FIGO grade 1 or 2 endometrioid adenocarcinoma. [6,7]
To explain this intriguing association it has been
hypothesized that some endometrial NECs may arise
from the neuroendocrine component of endometrioid
carcinomas. Although the possibility that an
[10]
abdominal NEC may secondarily develop due to
chemotherapy for an original endometrial carcinoma
should be mentioned, nevertheless in the present case
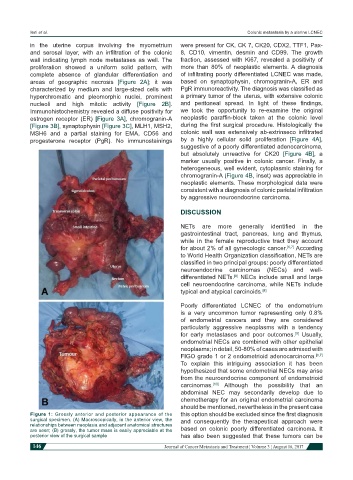
Figure 1: Grossly anterior and posterior appearance of the this option should be excluded since the first diagnosis
surgical specimen. (A) Macroscopically, in the anterior view, the and consequently the therapeutical approach were
relationships between neoplasia and adjacent anatomical structures
are seen; (B) grossly, the tumor mass is easily appreciable at the based on colonic poorly differentiated carcinoma. It
posterior view of the surgical sample has also been suggested that these tumors can be
146 Journal of Cancer Metastasis and Treatment ¦ Volume 3 ¦ August 16, 2017