Page 108 - Read Online
P. 108
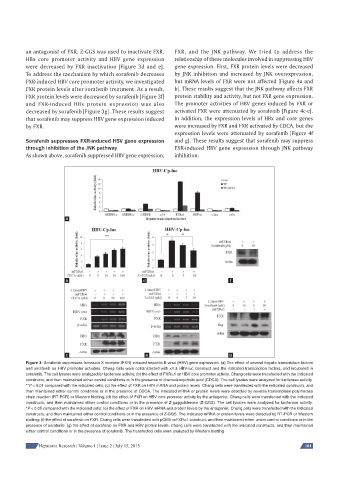
an antagonist of FXR, Z-GGS was used to inactivate FXR. FXR, and the JNK pathway. We tried to address the
HBx core promoter activity and HBV gene expression relationship of these molecules involved in suppressing HBV
were decreased by FXR inactivation [Figure 3d and e]. gene expression. First, FXR protein levels were decreased
To address the mechanism by which sorafenib decreases by JNK inhibition and increased by JNK overexpression,
FXR-induced HBV core promoter activity, we investigated but mRNA levels of FXR were not affected [Figure 4a and
FXR protein levels after sorafenib treatment. As a result, b]. These results suggest that the JNK pathway affects FXR
FXR protein levels were decreased by sorafenib [Figure 3f] protein stability and activity, but not FXR gene expression.
and FXR-induced HBx protein expression was also The promoter activities of HBV genes induced by FXR or
decreased by sorafenib [Figure 3g]. These results suggest activated FXR were attenuated by sorafenib [Figure 4c-e].
that sorafenib may suppress HBV gene expression induced In addition, the expression levels of HBx and core genes
by FXR. were increased by FXR and FXR activated by CDCA, but the
expression levels were attenuated by sorafenib [Figure 4f
Sorafenib suppresses FXR-induced HBV gene expression and g]. These results suggest that sorafenib may suppress
through inhibition of the JNK pathway FXR-induced HBV gene expression through JNK pathway
As shown above, sorafenib suppressed HBV gene expression, inhibition.
a
b d f
c e g
Figure 3: Sorafenib suppresses farnesoid X receptor (FXR)-induced hepatitis B virus (HBV) gene expression. (a) The effect of several hepatic transcription factors
and sorafenib on HBV promoter activities. Chang cells were cotransfected with ×1.3 HBV-luc construct and the indicated transcription factors, and incubated in
sorafenib. The cell lysates were analyzed for luciferase activity; (b) the effect of FXRα1 on HBV core promoter activity. Chang cells were transfected with the indicated
constructs, and then maintained either control conditions or in the presence of chenodeoxycholic acid (CDCA). The cell lysates were analyzed for luciferase activity.
**P < 0.01 compared with the indicated cells; (c) the effect of FXR on HBV mRNA and protein levels. Chang cells were transfected with the indicated constructs, and
then maintained either control conditions or in the presence of CDCA. The indicated mRNA or protein levels were detected by reverse transcriptase-polymerase
chain reaction (RT-PCR) or Western blotting; (d) the effect of FXR on HBV core promoter activity by the antagonist. Chang cells were transfected with the indicated
constructs, and then maintained either control conditions or in the presence of Z-guggulsterone (Z-GGS). The cell lysates were analyzed for luciferase activity.
*P < 0.05 compared with the indicated cells; (e) the effect of FXR on HBV mRNA and protein levels by the antagonist. Chang cells were transfected with the indicated
constructs, and then maintained either control conditions or in the presence of Z-GGS. The indicated mRNA or protein levels were detected by RT-PCR or Western
blotting; (f) the effect of sorafenib on FXR. Chang cells were transfected with pCMV-mFXRα1 construct, and then maintained either under control conditions or in the
presence of sorafenib; (g) the effect of sorafenib on FXR and HBV protein levels. Chang cells were transfected with the indicated constructs, and then maintained
either control conditions or in the presence of sorafenib. The transfected cells were analyzed by Western blotting
Hepatoma Research | Volume 1 | Issue 2 | July 15, 2015 101