Page 105 - Read Online
P. 105
Page 12 of 18 Yang et al. Energy Mater 2023;3:300029 https://dx.doi.org/10.20517/energymater.2023.10
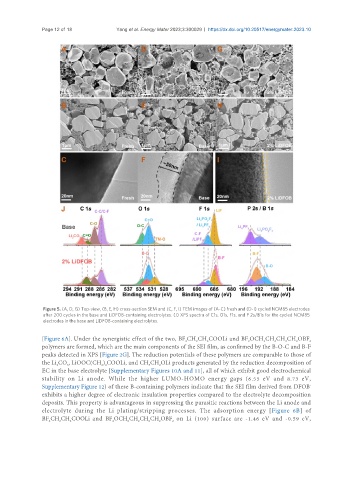
Figure 5. (A, D, G) Top-view, (B, E, H) cross-section SEM and (C, F, I) TEM images of (A-C) fresh and (D-I) cycled NCM85 electrodes
after 200 cycles in the base and LiDFOB-containing electrolytes. (J) XPS spectra of C1s, O1s, F1s, and P 2s/B1s for the cycled NCM85
electrodes in the base and LiDFOB-containing electrolytes.
[Figure 6A]. Under the synergistic effect of the two, BF CH CH COOLi and BF OCH CH CH CH OBF
2
2
2
2
2
2
2
2
2
polymers are formed, which are the main components of the SEI film, as confirmed by the B-O-C and B-F
peaks detected in XPS [Figure 2G]. The reduction potentials of these polymers are comparable to those of
the Li CO , LiOOC(CH ) COOLi, and CH CH OLi products generated by the reduction decomposition of
2 4
2
3
2
3
EC in the base electrolyte [Supplementary Figures 10A and 11], all of which exhibit good electrochemical
stability on Li anode. While the higher LUMO-HOMO energy gaps (6.55 eV and 8.73 eV,
Supplementary Figure 12) of these B-containing polymers indicate that the SEI film derived from DFOB
-
exhibits a higher degree of electronic insulation properties compared to the electrolyte decomposition
deposits. This property is advantageous in suppressing the parasitic reactions between the Li anode and
electrolyte during the Li plating/stripping processes. The adsorption energy [Figure 6B] of
BF CH CH COOLi and BF OCH CH CH CH OBF on Li (100) surface are -1.46 eV and -0.59 eV,
2
2
2
2
2
2
2
2
2