Page 107 - Read Online
P. 107
Page 14 of 18 Yang et al. Energy Mater 2023;3:300029 https://dx.doi.org/10.20517/energymater.2023.10
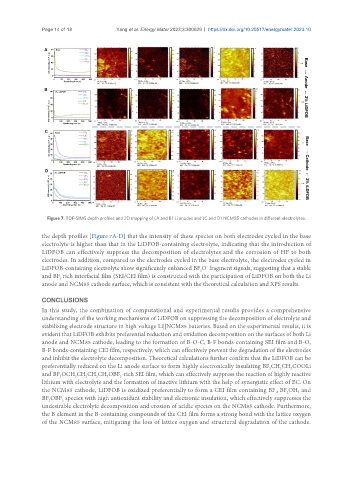
Figure 7. TOF-SIMS depth profiles and 2D mapping of (A and B) Li anodes and (C and D) NCM85 cathodes in different electrolytes.
the depth profiles [Figure 7A-D] that the intensity of these species on both electrodes cycled in the base
electrolyte is higher than that in the LiDFOB-containing electrolyte, indicating that the introduction of
LiDFOB can effectively suppress the decomposition of electrolytes and the corrosion of HF to both
electrodes. In addition, compared to the electrodes cycled in the base electrolyte, the electrodes cycled in
-
LiDFOB-containing electrolyte show significantly enhanced BF O fragment signals, suggesting that a stable
2
-
and BF rich interfacial film (SEI/CEI film) is constructed with the participation of LiDFOB on both the Li
2
anode and NCM85 cathode surface, which is consistent with the theoretical calculation and XPS results.
CONCLUSIONS
In this study, the combination of computational and experimental results provides a comprehensive
understanding of the working mechanisms of LiDFOB on suppressing the decomposition of electrolyte and
stabilizing electrode structure in high voltage Li||NCM85 batteries. Based on the experimental results, it is
evident that LiDFOB exhibits preferential reduction and oxidation decomposition on the surfaces of both Li
anode and NCM85 cathode, leading to the formation of B-O-C, B-F bonds-containing SEI film and B-O,
B-F bonds-containing CEI film, respectively, which can effectively prevent the degradation of the electrodes
and inhibit the electrolyte decomposition. Theoretical calculations further confirm that the LiDFOB can be
preferentially reduced on the Li anode surface to form highly electronically insulating BF CH CH COOLi
2
2
2
and BF OCH CH CH CH OBF -rich SEI film, which can effectively suppress the reaction of highly reactive
2
2
2
2
2
2
lithium with electrolyte and the formation of inactive lithium with the help of synergistic effect of EC. On
the NCM85 cathode, LiDFOB is oxidized preferentially to form a CEI film containing BF , BF OH, and
3
2
BF OBF species with high antioxidant stability and electronic insulation, which effectively suppresses the
2
2
undesirable electrolyte decomposition and erosion of acidic species on the NCM85 cathode. Furthermore,
the B element in the B-containing compounds of the CEI film forms a strong bond with the lattice oxygen
of the NCM85 surface, mitigating the loss of lattice oxygen and structural degradation of the cathode.