Page 10 - Read Online
P. 10
Page 6 of 14 Alvarez-Tirado et al. Energy Mater 2023;3:300003 https://dx.doi.org/10.20517/energymater.2022.59
Table 1. Iongel electrolyte compositions containing 90 wt.% liquid electrolyte and 10 wt.% PEGDM crosslinker
Liquid electrolyte [a]
Sample
IL Salt
Iongel-FSI* DEME-FSI LiTFSI in all cases
Iongel-TFSI* or DEME-TFSI
Iongel-TFSI
Iongel-FD* or FD-TFSI
Iongel-FD
Iongel-BETI* DEME-BETI
Iongel-CFSO* DEME-C F SO
3 9 3
Iongel-FSI DEME-FSI LiFSI
Iongel-BETI DEME-BETI LiBETI
Iongel-CFSO DEME-C F SO LiC F SO
3 9 3 3 9 3
[a]
At a 20 mol.% concentration of the selected salt. PEGDM: Poly(ethylene glycol) dimethacrylate.
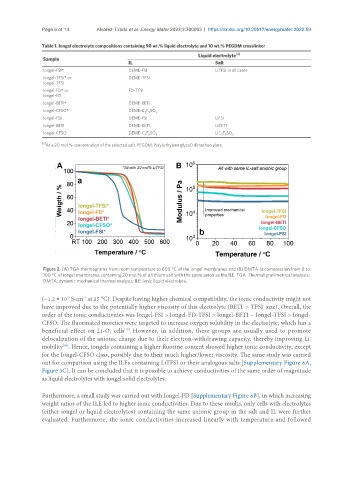
Figure 2. (A) TGA thermograms from room temperature to 600 °C of the iongel membranes and (B) DMTA at compression from 0 to
100 °C of iongel membranes containing 20 mol.% of a lithium salt with the same anion as the ILE. TGA: Thermal gravimetrical analysis;
DMTA: dynamic mechanical thermal analysis; ILE: ionic liquid electrolyte.
(~1.2 × 10 S·cm at 25 °C). Despite having higher chemical compatibility, the ionic conductivity might not
-3
-1
-
-
have improved due to the potentially higher viscosity of this electrolyte (BETI > TFSI size). Overall, the
order of the ionic conductivities was Iongel-FSI > Iongel-FD-TFSI > Iongel-BETI ~ Iongel-TFSI > Iongel-
CFSO. The fluorinated moieties were targeted to increase oxygen solubility in the electrolyte, which has a
beneficial effect on Li-O cells . However, in addition, these groups are usually used to promote
[17]
2
+
delocalization of the anionic charge due to their electron-withdrawing capacity, thereby improving Li
mobility . Hence, iongels containing a higher fluorine content showed higher ionic conductivity, except
[36]
for the Iongel-CFSO class, possibly due to their much higher/lower viscosity. The same study was carried
out for comparison using the ILEs containing LiTFSI or their analogous salts [Supplementary Figure 6A,
Figure 3C]. It can be concluded that it is possible to achieve conductivities of the same order of magnitude
as liquid electrolytes with iongel solid electrolytes.
Furthermore, a small study was carried out with Iongel-FD [Supplementary Figure 6B], in which increasing
weight ratios of the ILE led to higher ionic conductivities. Due to these results, only cells with electrolytes
(either iongel or liquid electrolytes) containing the same anionic group in the salt and IL were further
evaluated. Furthermore, the ionic conductivities increased linearly with temperature and followed