Page 13 - Read Online
P. 13
Alvarez-Tirado et al. Energy Mater 2023;3:300003 https://dx.doi.org/10.20517/energymater.2022.59 Page 9 of 14
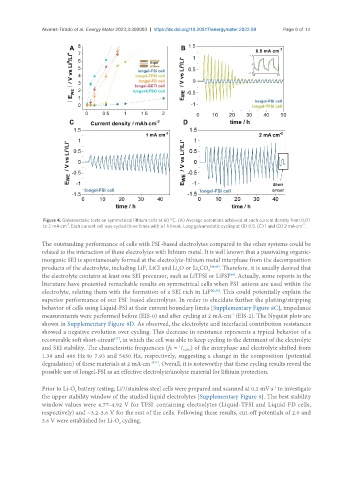
Figure 4. Galvanostatic tests on symmetrical lithium cells at 60 °C. (A) Average potentials achieved at each current density from 0.01
-2
-2
to 2 mA·cm . Each current cell was cycled three times with a 1 h break. Long galvanostatic cycling at (B) 0.5, (C) 1 and (D) 2 mA·cm .
The outstanding performance of cells with FSI -based electrolytes compared to the other systems could be
-
related to the interaction of these electrolytes with lithium metal. It is well known that a passivating organic-
inorganic SEI is spontaneously formed at the electrolyte-lithium metal interphase from the decomposition
products of the electrolyte, including LiF, LiCl and Li O or Li CO 3 [48,49] . Therefore, it is usually desired that
2
2
[48]
the electrolyte contains at least one SEI precursor, such as LiTFSI or LiFSI . Actually, some reports in the
literature have presented remarkable results on symmetrical cells when FSI anions are used within the
-
electrolyte, relating them with the formation of a SEI rich in LiF [42,50] . This could potentially explain the
superior performance of our FSI based electrolytes. In order to elucidate further the platting/stripping
-
behavior of cells using Liquid-FSI at their current boundary limits [Supplementary Figure 8C], impedance
measurements were performed before (EIS-0) and after cycling at 2 mA·cm (EIS-2). The Nyquist plots are
-2
shown in Supplementary Figure 8D. As observed, the electrolyte and interfacial contribution resistances
showed a negative evolution over cycling. This decrease in resistance represents a typical behavior of a
recoverable soft short-circuit , in which the cell was able to keep cycling to the detriment of the electrolyte
[47]
and SEI stability. The characteristic frequencies (fc = / 2πRC ) of the interphase and electrolyte shifted from
1
1.38 and 486 Hz to 7.93 and 5430 Hz, respectively, suggesting a change in the composition (potential
degradation) of these materials at 2 mA·cm -2[51] . Overall, it is noteworthy that these cycling results reveal the
possible use of Iongel-FSI as an effective electrolyte/anolyte material for lithium protection.
Prior to Li-O battery testing, Li //stainless steel cells were prepared and scanned at 0.2 mV·s to investigate
-1
0
2
the upper stability window of the studied liquid electrolytes [Supplementary Figure 9]. The best stability
window values were 4.77-4.92 V for TFSI containing electrolytes (Liquid-TFSI and Liquid-FD cells,
-
respectively) and ~3.2-3.6 V for the rest of the cells. Following these results, cut-off potentials of 2.0 and
3.6 V were established for Li-O cycling.
2