Page 14 - Read Online
P. 14
Page 10 of 14 Alvarez-Tirado et al. Energy Mater 2023;3:300003 https://dx.doi.org/10.20517/energymater.2022.59
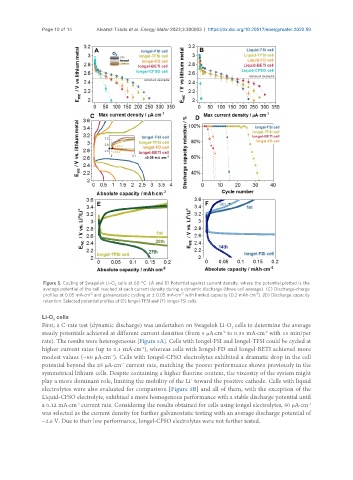
Figure 5. Cycling of Swagelok Li-O cells at 60 °C. (A and B) Potential against current density, where the potential plotted is the
2
average potential of the cell reached at each current density during a dynamic discharge (three cell average). (C) Discharge-charge
-2
-2
-2
profiles at 0.05 mA·cm and galvanostatic cycling at ± 0.05 mA·cm with limited capacity (0.2 mAh·cm ). (D) Discharge capacity
retention. Selected potential profiles of (E) Iongel-TFSI and (F) Iongel-FSI cells.
Li-O cells
2
First, a C-rate test (dynamic discharge) was undertaken on Swagelok Li-O cells to determine the average
2
steady potentials achieved at different current densities (from 5 μA·cm to 0.35 mA·cm with 15 min/per
-2
-2
rate). The results were heterogeneous [Figure 5A]. Cells with Iongel-FSI and Iongel-TFSI could be cycled at
higher current rates (up to 0.1 mA·cm ), whereas cells with Iongel-FD and Iongel-BETI achieved more
-2
modest values (~60 μA·cm ). Cells with Iongel-CFSO electrolytes exhibited a dramatic drop in the cell
-2
-2
potential beyond the 25 μA·cm current rate, matching the poorer performance shown previously in the
symmetrical lithium cells. Despite containing a higher fluorine content, the viscosity of the system might
+
play a more dominant role, limiting the mobility of the Li toward the positive cathode. Cells with liquid
electrolytes were also evaluated for comparison [Figure 5B] and all of them, with the exception of the
Liquid-CFSO electrolyte, exhibited a more homogenous performance with a stable discharge potential until
-2
a 0.12 mA·cm current rate. Considering the results obtained for cells using iongel electrolytes, 50 μA·cm
-2
was selected as the current density for further galvanostatic testing with an average discharge potential of
~2.6 V. Due to their low performance, Iongel-CFSO electrolytes were not further tested.