Page 11 - Read Online
P. 11
Alvarez-Tirado et al. Energy Mater 2023;3:300003 https://dx.doi.org/10.20517/energymater.2022.59 Page 7 of 14
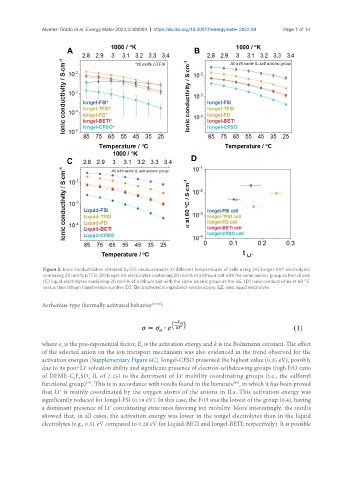
Figure 3. Ionic conductivities obtained by EIS measurements at different temperatures of cells using (A) Iongel-XX* electrolytes
containing 20 mol.% LiTFSI, (B) Iongel-XX electrolytes containing 20 mol.% of a lithium salt with the same anionic group as the ILE and
(C) liquid electrolytes containing 20 mol.% of a lithium salt with the same anionic group as the ILE. (D) Ionic conductivities at 60 °C
versus their lithium transference number. EIS: Electrochemical impedance spectroscopy; ILE: ionic liquid electrolyte.
Arrhenius-type thermally activated behavior [37,38] :
where σ is the pre-exponential factor, E is the activation energy and k is the Boltzmann constant. The effect
o
a
of the selected anion on the ion transport mechanism was also evidenced in the trend observed for the
activation energies [Supplementary Figure 6C]. Iongel-CFSO presented the highest value (0.35 eV), possibly
due to its poor Li solvation ability and significant presence of electron-withdrawing groups (high F/O ratio
+
of DEME-C F SO IL of 2.25) to the detriment of Li mobility coordinating groups (i.e., the sulfonyl
+
3 9
3
functional group) . This is in accordance with results found in the literature , in which it has been proved
[40]
[39]
+
that Li is mainly coordinated by the oxygen atoms of the anions in ILs. This activation energy was
significantly reduced for Iongel-FSI (0.18 eV). In this case, the F/O was the lowest of the group (0.4), having
a dominant presence of Li coordinating structures favoring ion mobility. More interestingly, the results
+
showed that, in all cases, the activation energy was lower in the iongel electrolytes than in the liquid
electrolytes (e.g., 0.31 eV compared to 0.28 eV for Liquid-BETI and Iongel-BETI, respectively). It is possible