Page 120 - Read Online
P. 120
Li et al. Microstructures 2023;3:2023024 https://dx.doi.org/10.20517/microstructures.2023.09 Page 9 of 20
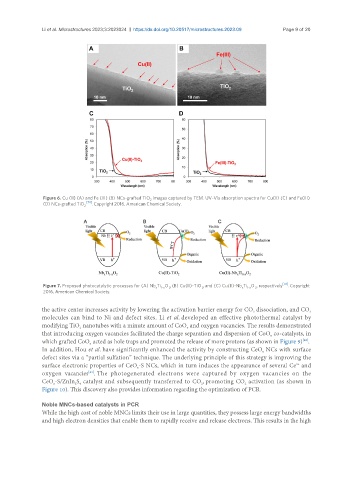
Figure 6. Cu (II) (A) and Fe (III) (B) NCs-grafted TiO images captured by TEM. UV-Vis absorption spectra for Cu(II) (C) and Fe(III)
2
(D) NCs-grafted TiO 2 [70] . Copyright 2016, American Chemical Society.
[70]
Figure 7. Proposed photocatalytic processes for (A) Nb Ti O , (B) Cu(II)-TiO , and (C) Cu(II)-Nb Ti O , respectively . Copyright
x 1-x 2 2 x 1-x 2
2016, American Chemical Society.
the active center increases activity by lowering the activation barrier energy for CO dissociation, and CO
2
2
molecules can bind to Ni and defect sites. Li et al. developed an effective photothermal catalyst by
modifying TiO nanotubes with a minute amount of CoO and oxygen vacancies. The results demonstrated
x
2
that introducing oxygen vacancies facilitated the charge separation and dispersion of CoO co-catalysts, in
x
[82]
which grafted CoO acted as hole traps and promoted the release of more protons (as shown in Figure 9) .
x
In addition, Hou et al. have significantly enhanced the activity by constructing CeO NCs with surface
x
defect sites via a “partial sulfation” technique. The underlying principle of this strategy is improving the
surface electronic properties of CeO -S NCs, which in turn induces the appearance of several Ce and
3+
x
oxygen vacancies . The photogenerated electrons were captured by oxygen vacancies on the
[83]
CeO -S/ZnIn S catalyst and subsequently transferred to CO , promoting CO activation (as shown in
x
2 4
2
2
Figure 10). This discovery also provides information regarding the optimization of PCR.
Noble MNCs-based catalysts in PCR
While the high cost of noble MNCs limits their use in large quantities, they possess large energy bandwidths
and high electron densities that enable them to rapidly receive and release electrons. This results in the high