Page 251 - Read Online
P. 251
Rice et al. Minocycline in spinal cord injury
the over-activation of which contributes to neural cell
death, minocycline reduces toxicity to oligodendrocytes
and neurons through potentially direct mechanisms.
In this regard, minocycline inhibits the activity of
caspases [21,22] and the release of cytochrome c from
mitochondria [23] , which are both apoptosis-inducing
events. Minocycline reduces signaling of the p38
mitogen activated kinase pathway [6,24] , and it prevents
the activation of poly (ADP-ribose) polymerase [25,26] ,
actions that contribute to the alleviation of neural cell
death. This drug has also been shown to decrease
apoptosis of oligodendrocytes through a mechanism
involving the inhibition of proNGF production by
microglia [27] . Minocycline has been reported to reduce
glutamate excitotoxicity [28,29] , to detoxify free radicals
that contribute to neurotoxicity [30,31] and to inhibit lipid
peroxidation [32] .
A limitation of the present study is that we did not
perform neurobehavioral studies to accompany
the histological and MMP results. However, the
dose regimen employed is identical to that used in
our previous study that demonstrated behavioral
[5]
recovery by 3 days post-injury in the minocycline
compared to vehicle group. Another limitation is that
for the majority of MMPs examined in the current
study, only gene expression and not protein amount
or activity was measured. However, protein levels of
MMPs-2 and 9 were measured using gel zymography,
and net proteolytic activity for these two molecules
were examined with in situ zymography. Despite this
limitation, the transcript expression pattern of MMPs
provides valuable information in understanding the
mechanisms by which minocycline may exert its
effects after SCI.
In conclusion, the novel findings are that minocycline
confers protection to neurons at the site of SCI,
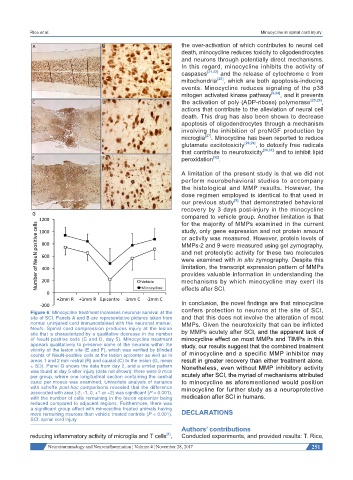
Figure 6: Minocycline treatment increases neuronal survival at the
site of SCI. Panels A and B are representative pictures taken from and that this does not involve the alteration of most
normal uninjured cord immunostained with the neuronal marker, MMPs. Given the neurotoxicity that can be inflicted
NeuN. Spinal cord compression produces injury at the lesion
site that is characterized by a qualitative decrease in the number by MMPs acutely after SCI, and the apparent lack of
of NeuN positive cells (C and D, day 5). Minocycline treatment minocycline effect on most MMPs and TIMPs in this
appears qualitatively to preserve some of the neurons within the study, our results suggest that the combined treatment
vicinity of the lesion site (E and F), which was verified by blinded
counts of NeuN-positive cells at the lesion epicenter as well as in of minocycline and a specific MMP inhibitor may
areas 1 and 2 mm rostral (R) and caudal (C) to the lesion (G, mean result in greater recovery than either treatment alone.
± SD). Panel G shows the data from day 2, and a similar pattern Nonetheless, even without MMP inhibitory activity
was found at day 5 after injury (data not shown); there were 9 mice
per group, where one longitudinal section containing the central acutely after SCI, the myriad of mechanisms attributed
canal per mouse was examined. Univariate analysis of variance to minocycline as aforementioned would position
with scheffe post-hoc comparisons revealed that the difference minocycline for further study as a neuroprotective
associated with area (-2, -1, 0, +1 or +2) was significant (P < 0.001),
with the number of cells remaining in the lesion epicenter being medication after SCI in humans.
reduced compared to adjacent regions. Furthermore, there was
a significant group effect with minocycline treated animals having
more remaining neurons than vehicle treated controls (P < 0.001). DECLARATIONS
SCI: spinal cord injury
Authors’ contributions
[1]
reducing inflammatory activity of microglia and T cells , Conducted experiments, and provided results: T. Rice,
Neuroimmunology and Neuroinflammation ¦ Volume 4 ¦ November 28, 2017 251