Page 24 - Read Online
P. 24
is challenging. It varies 0.1‑2% in all ischemic strokes and
4‑18% in thalamic infarction. Here, we report a case of
[2]
bilateral thalamic infarct due to occlusion of the AOP.
CASE REPORT
A 79‑year‑old right‑handed Parkinsonian female was
found unresponsive in her bed at home. She was last
seen normal approximately 8 h prior to her admission.
There was no recent history of fever, headache, seizure,
trauma, and known exposure to toxic substances.
There was no history of any memory impairment or
dementia. On examination, the patient was drowsy with
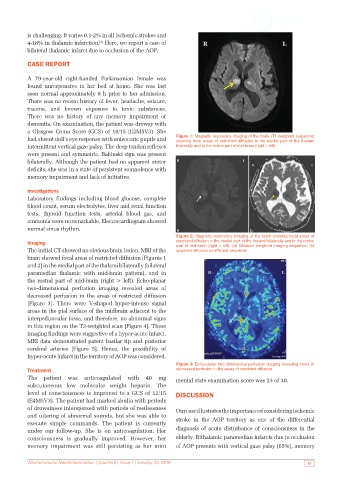
a Glasgow Coma Score (GCS) of 10/15 (E2M5V3). She
had absent doll’s eye response with anisocoric pupils and Figure 1: Magnetic resonance imaging of the brain (T1-weighted sequence)
showing focal areas of restricted diffusion in the medial part of the thalami
intermittent vertical gaze palsy. The deep tendon reflexes bilaterally and in the rostral part of mid‑brain (right > left)
were present and symmetric. Babinski sign was present
bilaterally. Although the patient had no apparent motor
deficits, she was in a state of persistent somnolence with
memory impairment and lack of initiative.
Investigations
Laboratory findings including blood glucose, complete
blood count, serum electrolytes, liver and renal function
tests, thyroid function tests, arterial blood gas, and
ammonia were unremarkable. Electrocardiogram showed
normal sinus rhythm.
Figure 2: Magnetic resonance imaging of the brain showing focal areas of
Imaging restricted diffusion in the medial part of the thalami bilaterally and in the rostral
part of mid‑brain (right > left). (a) Diffusion‑weighted imaging sequence; (b)
The initial CT showed no obvious brain lesion. MRI of the apparent diffusion co-efficient sequence
brain showed focal areas of restricted diffusion [Figures 1
and 2] in the medial part of the thalami bilaterally (bilateral
paramedian thalamic with mid‑brain pattern), and in
the rostral part of mid‑brain (right > left). Echo‑planar
two‑dimensional perfusion imaging revealed areas of
decreased perfusion in the areas of restricted diffusion
[Figure 3]. There were V‑shaped hyper‑intense signal
areas in the pial surface of the midbrain adjacent to the
interpeduncular fossa, and therefore, no abnormal signs
in this region on the T2‑weighted scan [Figure 4]. These
imaging findings were suggestive of a hyper‑acute infarct.
MRI data demonstrated patent basilar tip and posterior
cerebral arteries [Figure 5]. Hence, the possibility of
hyper‑acute infarct in the territory of AOP was considered.
Figure 3: Echo‑planar two‑dimensional perfusion imaging revealing areas of
Treatment decreased perfusion in the areas of restricted diffusion
The patient was anticoagulated with 40 mg mental state examination score was 23 of 30.
subcutaneous low molecular weight heparin. The
level of consciousness is improved to a GCS of 12/15 DISCUSSION
(E4M5V3). The patient had marked abulia with periods
of drowsiness interspersed with periods of restlessness Our case illustrates the importance of considering ischemic
and uttering of abnormal sounds, but she was able to stroke in the AOP territory as one of the differential
execute simple commands. The patient is currently
under our follow‑up. She is on anticoagulation. Her diagnosis of acute disturbance of consciousness in the
consciousness is gradually improved. However, her elderly. Bithalamic paramedian infarcts due to occlusion
memory impairment was still persisting as her mini of AOP presents with vertical gaze palsy (65%), memory
Neuroimmunol Neuroinfammation | Volume 3 | Issue 1 | January 20, 2016 15