Page 265 - Read Online
P. 265
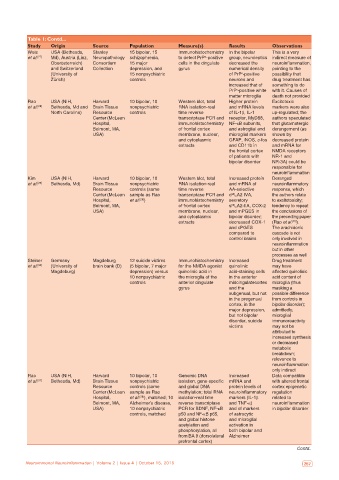
Table 1: Contd...
Study Origin Source Population Measure(s) Results Observations
Weis USA (Bethesda, Stanley 15 bipolar, 15 Immunohistochemistry In the bipolar This is a very
et al. [27] Md), Austria (Linz, Neuropathology schizophrenia, to detect PrP ‑positive group, neuroleptics indirect measure of
c
Oberösterreich) Consortium 15 major cells in the cingulate decreased the neuroinflammation,
and Switzerland Collection depression, and gyrus numerical density pointing to the
c
(University of 15 nonpsychiatric of PrP ‑positive possibility that
Zürich) controls neurons and drug treatment has
increased that of something to do
PrP ‑positive white with it. Causes of
c
matter microglia death not provided
Rao USA (NIH, Harvard 10 bipolar, 10 Western blot, total Higher protein Excitotoxic
et al. [28] Bethesda, Md and Brain Tissue nonpsychiatric RNA isolation‑real and mRNA levels markers were also
North Carolina) Resource controls time reverse of IL‑1β, IL‑1 up‑regulated; the
Center (McLean transcriptase PCR and receptor, MyD88, authors speculated
Hospital, immunohistochemistry NF‑κB subunits, that glutamatergic
Belmont, MA, of frontal cortex and astroglial and derangement (as
USA) membrane, nuclear, microglial markers shown by
and cytoplasmic GFAP, iNOS, c‑fos decreased protein
extracts and CD11b in and mRNA for
the frontal cortex NMDA receptors
of patients with NR‑1 and
bipolar disorder NR‑3A) could be
responsible for
neuroinflammation
Kim USA (NIH, Harvard 10 bipolar, 10 Western blot, total Increased protein Deranged
et al. [29] Bethesda, Md) Brain Tissue nonpsychiatric RNA isolation‑real and mRNA of neuroinflammatory
Resource controls (same time reverse AA‑selective response, which
Center (McLean sample as Rao transcriptase PCR and cPLA2 IVA, the authors relate
Hospital, et al. ) immunohistochemistry secretory to excitotoxicity;
[28]
Belmont, MA, of frontal cortex sPLA2‑IIA, COX‑2 tendency to repeat
USA) membrane, nuclear, and mPGES in the conclusions of
and cytoplasmic bipolar disorder; the preceding paper
extracts decreased COX‑1 (Rao et al. ).
[28]
and cPGES The arachidonic
compared to cascade is not
control brains only involved in
neuroinflammation
but in other
processes as well
Steiner Germany Magdeburg 12 suicide victims Immunohistochemistry Increased Drug treatment
et al. [30] (University of brain bank (D) (5 bipolar, 7 major for the NMDA agonist quinolinic may have
Magdeburg) depression) versus quinolinic acid in acid‑staining cells affected quinolinic
10 nonpsychiatric the microglia of the in the anterior acid content of
controls anterior cingulate midcingulatecortex microglia (thus
gyrus and the masking a
subgenual, but not possible difference
in the pregenual from controls in
cortex, in the bipolar disorder);
major depression, admittedly,
but not bipolar microglial
disorder, suicide immunoreactivity
victims may not be
attributed to
increased synthesis
or decreased
metabolic
breakdown;
relevance to
neuroinflammation
only indirect
Rao USA (NIH, Harvard 10 bipolar, 10 Genomic DNA Increased Data compatible
et al. [31] Bethesda, Md) Brain Tissue nonpsychiatric isolation, gene‑specific mRNA and with altered frontal
Resource controls (same and global DNA protein levels of cortex epigenetic
Center (McLean sample as Rao methylation; total RNA neuroinflammatory regulation
Hospital, et al. ), matched; 10 isolation‑real time markers (IL‑1β related to
[28]
Belmont, MA, Alzheimer’s disease, reverse transcriptase and TNF‑α) neuroinflammation
USA) 10 nonpsychiatric PCR for BDNF, NF‑κB and of markers in bipolar disorder
controls, matched p50 and NF‑κB p65, of astrocytic
and global histone and microglial
acetylation and activation in
phosphorylation, all both bipolar and
from BA 9 (dorsolateral Alzheimer
prefrontal cortex)
Contd..
Neuroimmunol Neuroinflammation | Volume 2 | Issue 4 | October 15, 2015 257