Page 185 - Read Online
P. 185
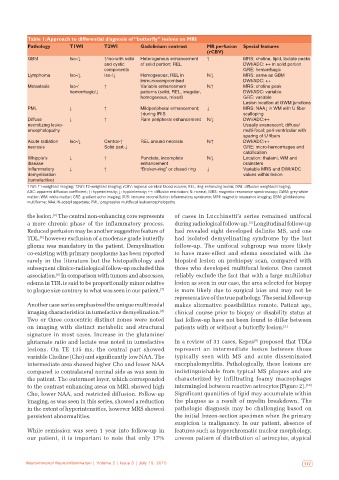
Table 1: Approach to differential diagnosis of “butterfly” lesions on MRI
Pathology T1WI T2WI Gadolinium contrast MR perfusion Special features
(rCBV)
GBM Iso‑/↓ ↑/Iso‑with solid Heterogenous enhancement ↑ MRS: choline, lipid, lactate peaks
and cystic of solid portion; REL DWI/ADC: ++ in solid portion
components GRE: hemorrhagic
Lymphoma Iso‑/↓ Iso‑/↓ Homogenous; REL in N/↓ MRS: same as GBM
immunocompromised DWI/ADC: ++
Metastasis Iso‑/ ↑ Variable enhancement N/↑ MRS: choline peak
hemorrhagic/↓ patterns (solid, REL, irregular, DWI/ADC: variable
homogenous, mixed) GRE: variable
Lesion location at GWM junctions
PML ↓ ↑ Mildperipheral enhancement; ↓ MRS: NAA↓ in WM with U fiber
↑during IRIS scalloping
Diffuse ↓ ↑ Rare peripheral enhancement N/↓ DWI/ADC:++
necrotizing leuko‑ Usually evanescent, diffuse/
encephalopathy multi‑focal; peri‑ventricular with
sparing of U fibers
Acute radiation Iso‑/↓ Central‑↑ REL around necrosis N/↑ DWI/ADC:++
necrosis Solid part‑↓ GRE: micro‑hemorrhages and
calcification
Whipple’s ↓ ↑ Punctate, incomplete N/↓ Location: thalami, WM and
disease enhancement brainstem
Inflammatory ↓ ↑ “Broken‑ring” or closed ring ↓ Variable MRS and DWI/ADC
demyelination values within lesion
(tumefactive)
T1WI: T1‑weighted imaging; T2WI: T2‑weighted imaging; rCBV: regional cerebral blood volume; REL: ring‑enhancing lesion; DWI: diffusion weighted imaging;
ADC: apparent diffusion coefficient; ↑: hyperintensity; ↓: hypointensity; ++: diffusion restriction; N: normal; MRS: magnetic resonance spectroscopy; GWM: grey‑white
matter; WM: white matter; GRE: gradient echo imaging; IRIS: immune reconstitution inflammatory syndrome; MRI: magnetic resonance imaging; GBM: glioblastoma
multiforme; NAA: N‑acetyl aspartate; PML: progressive multifocal leukoencephalopathy
the lesion. The central non-enhancing core represents of cases in Lucchinetti’s series remained unifocal
[2]
[3]
a more chronic phase of the inflammatory process. during radiological follow-up. Longitudinal follow-up
Reduced perfusion may be another suggestive feature of had revealed eight developed definite MS, and one
TDL, however exclusion of a moderate grade butterfly had isolated demyelinating syndrome by the last
[5]
glioma was mandatory in the patient. Demyelination follow-up. The unifocal subgroup was more likely
co-existing with primary neoplasms has been reported to have mass-effect and edema associated with the
rarely in the literature but the histopathology and biopsied lesion on prebiopsy scan, compared with
subsequent clinico-radiological follow-up excluded this those who developed multifocal lesions. One cannot
association. In comparison with tumors and abscesses, reliably exclude the fact that with a large multilobar
[6]
edema in TDL is said to be proportionally minor relative lesion as seen in our case, the area selected for biopsy
to plaque size contrary to what was seen in our patient. [7] is more likely due to surgical bias and may not be
representative of the true pathology. The serial follow-up
Another case series emphasized the unique multimodal makes alternative possibilities remote. Patient age,
imaging characteristics in tumefactive demyelination. clinical course prior to biopsy or disability status at
[8]
Two or three concentric distinct zones were noted last follow-up have not been found to differ between
on imaging with distinct metabolic and structural patients with or without a butterfly lesion. [3]
signature in most cases. Increase in the glutamine/
[9]
glutamate ratio and lactate was noted in tumefactive In a review of 31 cases, Kepes proposed that TDLs
lesions. On TE 135 ms, the central part showed represent an intermediate lesion between those
variable Choline (Cho) and significantly low NAA. The typically seen with MS and acute disseminated
intermediate area showed higher Cho and lower NAA encephalomyelitis. Pathologically, these lesions are
compared to contralateral normal side as was seen in indistinguishable from typical MS plaques and are
the patient. The outermost layer, which corresponded characterized by infiltrating foamy macrophages
to the contrast enhancing areas on MRI, showed high intermingled between reactive astrocytes [Figure 2]. [10]
Cho, lower NAA, and restricted diffusion. Follow-up Significant quantities of lipid may accumulate within
imaging, as was seen in this series, showed a reduction the plaques as a result of myelin breakdown. The
in the extent of hyperintensities, however MRS showed pathologic diagnosis may be challenging based on
persistent abnormalities. the initial frozen-section specimen when the primary
suspicion is malignancy. In our patient, absence of
While remission was seen 1 year into follow-up in features such as hyperchromatic nuclear morphology,
our patient, it is important to note that only 17% uneven pattern of distribution of astrocytes, atypical
176 Neuroimmunol Neuroinflammation | Volume 2 | Issue 3 | July 15, 2015 Neuroimmunol Neuroinflammation | Volume 2 | Issue 3 | July 15, 2015 177