Page 25 - Read Online
P. 25
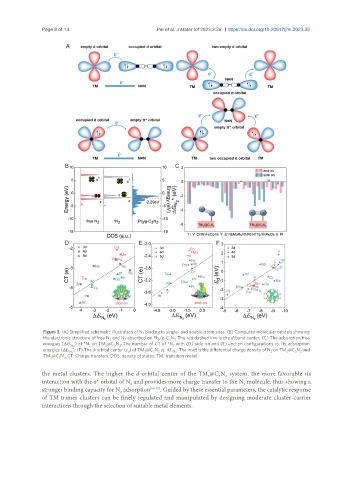
Page 8 of 14 Pei et al. J Mater Inf 2023;3:26 https://dx.doi.org/10.20517/jmi.2023.35
Figure 3. (A) Simplified schematic illustration of N binding to single- and double-atom sites; (B) Computed molecular orbitals showing
2
the electronic structure of free N and N absorbed on Pt /g-C N . The red dashed line is the d-band center; (C) The adsorption free
2 2 3 3 3
energies (ΔG ) of *N on TM @C N . The number of CT of *N with (D) side-on and (E) end-on configurations vs. its adsorption
*N 2 2 3 3 3 2
. The inset is the differential charge density of N on TM @C N and
d
3
3
3
energies (ΔE ); (F) The d-orbital center (ε ) of TM @C N vs. ΔE *N 2 2 3 3 3
*N 2
TM @C N . CT: Charge transfers; DOS: density of states; TM: transition metal.
3
3
3
the metal clusters. The higher the d orbital center of the TM @C N system, the more favorable its
3
3
3
interaction with the π* orbital of N and provides more charge transfer to the N molecule, thus showing a
2
2
stronger binding capacity for N adsorption [69-72] . Guided by these essential parameters, the catalytic response
2
of TM trimer clusters can be finely regulated and manipulated by designing moderate cluster-carrier
interactions through the selection of suitable metal elements.