Page 27 - Read Online
P. 27
Page 10 of 14 Pei et al. J Mater Inf 2023;3:26 https://dx.doi.org/10.20517/jmi.2023.35
Table 2. DFT-calculated potential-determining steps (including reaction mechanisms and corresponding reactions) and
corresponding limiting potentials (U ) for TM @C N (TM = Re, Pt, Ru, Rh, Ta, Ir)
3
L
3
3
Potential-determining step
TM @C N 3 Mechanism Reaction U (V)
3
3
L
-
+
Re Consecutive *HNNH + H + e → *HNNH 2 -0.11
+ -
Pt Enzymatic *NH + H + e → *NH -0.24
2 3
+ -
Ru Enzymatic *N + H + e → *NNH -0.35
2 + -
Consecutive *NNH+ H + e → *NNH 2 -0.40
+ -
Rh Enzymatic *N + H + e → *NNH -0.40
2
Consecutive
+
-
Ta Consecutive *HNNH + H + e → *HNNH 2 -0.42
+ -
Ir Enzymatic *NH + H + e → *NH -0.54
2 3
Consecutive
DFT: Density functional theory; TM: transition metal.
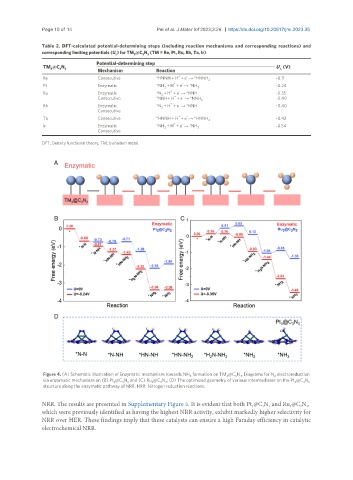
Figure 4. (A) Schematic illustration of Enzymatic mechanism towards NH formation on TM @C N . Diagrams for N electroreduction
3 3 3 3 2
via enzymatic mechanism on (B) Pt @C N and (C) Ru @C N ; (D) The optimized geometry of various intermediates on the Pt @C N 3
3
3
3
3
3
3
3
3
structure along the enzymatic pathway of NRR. NRR: Nitrogen reduction reactions.
NRR. The results are presented in Supplementary Figure 3. It is evident that both Pt @C N and Ru @C N ,
3
3
3
3
3
3
which were previously identified as having the highest NRR activity, exhibit markedly higher selectivity for
NRR over HER. These findings imply that these catalysts can ensure a high Faraday efficiency in catalytic
electrochemical NRR.