Page 31 - Read Online
P. 31
Page 6 of 23 Shanmugasundaram et al. Energy Mater. 2025, 5, 500100 https://dx.doi.org/10.20517/energymater.2024.304
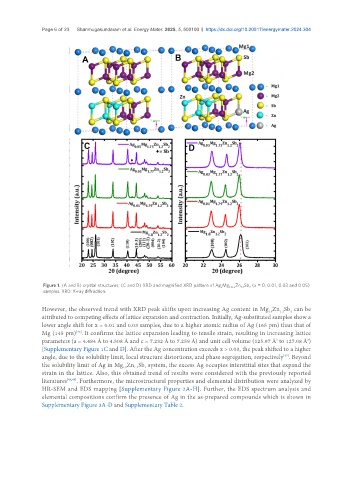
Figure 1. (A and B) crystal structures; (C and D) XRD and magnified XRD pattern of Ag Mg 1.8-x Zn Sb (x = 0, 0.01, 0.03 and 0.05)
x
1.2
2
samples. XRD: X-ray diffraction.
However, the observed trend with XRD peak shifts upon increasing Ag content in Mg Zn Sb can be
2
1.2
1.8
attributed to competing effects of lattice expansion and contraction. Initially, Ag-substituted samples show a
lower angle shift for x = 0.01 and 0.03 samples, due to a higher atomic radius of Ag (165 pm) than that of
Mg (145 pm) . It confirms the lattice expansion leading to tensile strain, resulting in increasing lattice
[45]
parameters (a = 4.484 Å to 4.508 Å and c = 7.232 Å to 7.259 Å) and unit cell volume (125.97 Å to 127.08 Å )
3
3
[Supplementary Figure 1C and D]. After the Ag concentration exceeds x > 0.03, the peak shifted to a higher
angle, due to the solubility limit, local structure distortions, and phase segregation, respectively . Beyond
[47]
the solubility limit of Ag in Mg Zn Sb system, the excess Ag occupies interstitial sites that expand the
1.8
1.2
2
strain in the lattice. Also, this obtained trend of results were considered with the previously reported
literatures [48,49] . Furthermore, the microstructural properties and elemental distribution were analyzed by
HR-SEM and EDS mapping [Supplementary Figure 2A-H]. Further, the EDS spectrum analysis and
elemental compositions confirm the presence of Ag in the as-prepared compounds which is shown in
Supplementary Figure 3A-D and Supplementary Table 2.