Page 238 - Read Online
P. 238
Page 8 of 33 Girase et al. Energy Mater. 2025, 5, 500132 https://dx.doi.org/10.20517/energymater.2025.14
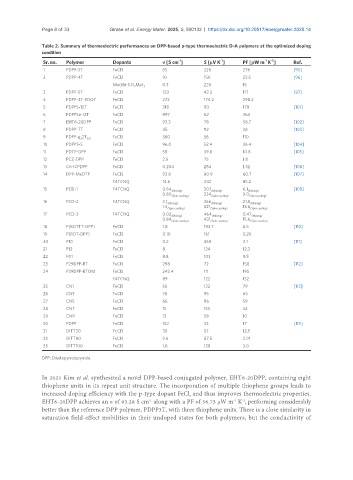
Table 2. Summary of thermoelectric performances on DPP-based p-type thermoelectric D-A polymers at the optimized doping
condition
-1
Sr. no. Polymer Dopants σ [S cm ] S [μV K ] PF [μW m K ] Ref.
-1
-2
-1
1 PDPP-3T FeCl3 55 226 276 [95]
2 PDPP-4T FeCl3 10 150 23.5 [96]
Mo(tfd-CO Me) 3 0.3 220 15
2
3 PDPP-5T FeCl3 120 42.2 11.1 [97]
4 PDPP-4T-EDOT FeCl3 272 174.2 298.2
5 PDPPS-12T FeCl3 318 80 178 [101]
6 PDPPSe-12T FeCl3 997 62 364
7 EHT6-20DPP FeCl3 93.3 78 56.7 [102]
8 PDPP-TT FeCl3 45 92 38 [103]
9 PDPP-g 2T 0.3 FeCl3 360 56 110
3
10 PDPPS-5 FeCl3 96.0 52.4 26.4 [104]
11 PDTP-DPP FeCl3 58 39.8 10.8 [105]
12 PCZ-DPP FeCl3 2.5 75 1.8
13 C6-ICPDPP FeCl3 0.204 254 1.32 [106]
14 DPP-MeDTP FeCl3 92.8 80.9 60.7 [107]
F4TCNQ 14.6 242 85.2
15 PCD-1 F4TCNQ 0.64 (Mixing) 307 (Mixing) 6.1 (Mixing) [108]
0.80 334 9.0
(Spin coating) (Spin coating) (Spin coating)
16 PCD-2 F4TCNQ 3.1 (Mixing) 266 (Mixing) 21.8 (Mixing)
1.3 321 13.6
(Spin coating) (Spin coating) (Spin coating)
17 PCD-3 F4TCNQ 0.02 (Mixing) 464 (Mixing) 0.47 (Mixing)
0.84 431 15.6
(Spin coating) (Spin coating) (Spin coating)
18 P(BDTTT-DPP) FeCl3 1.8 193.7 6.5 [110]
19 P(BDT-DPP) FeCl3 0.10 161 0.26
20 P10 FeCl3 0.2 360 3.1 [111]
21 P12 FeCl3 8 124 12.3
22 P01 FeCl3 8.8 103 9.5
23 P29DPP-BT FeCl3 298 72 158 [112]
24 P29DPP-BTOM FeCl3 242.4 111 195
F4TCNQ 89 122 132
25 CN1 FeCl3 56 122 79 [113]
26 CN3 FeCl3 78 95 65
27 CN5 FeCl3 66 96 59
28 CN7 FeCl3 15 155 34
29 CN9 FeCl3 13 58 10
30 PDPP FeCl3 152 33 17 [119]
31 DITT30 FeCl3 38 57 12.5
32 DITT80 FeCl3 2.6 87.5 2.01
33 DITT100 FeCl3 1.6 138 3.0
DPP: Diketopyrrolopyrrole.
In 2021 Kim et al. synthesized a novel DPP-based conjugated polymer, EHT6-20DPP, containing eight
thiophene units in its repeat unit structure. The incorporation of multiple thiophene groups leads to
increased doping efficiency with the p-type dopant FeCl and thus improves thermoelectric properties.
3
EHT6-20DPP achieves an σ of 93.28 S cm along with a PF of 56.73 μW m K , performing considerably
-2
-1
-1
better than the reference DPP polymer, PDPP3T, with three thiophene units. There is a close similarity in
saturation field-effect mobilities in their undoped states for both polymers, but the conductivity of