Page 237 - Read Online
P. 237
Girase et al. Energy Mater. 2025, 5, 500132 https://dx.doi.org/10.20517/energymater.2025.14 Page 7 of 33
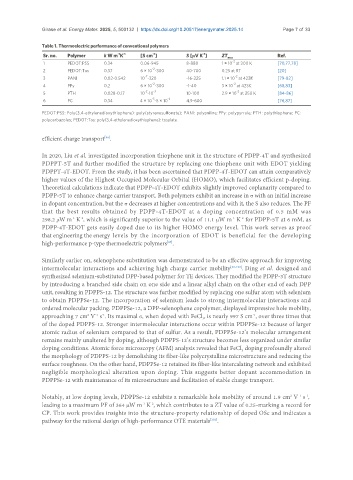
Table 1. Thermoelectric performance of conventional polymers
Sr. no. Polymer k W m K -1 [S cm ] S [μV K ] ZT max Ref.
-1
-1
-1
-2
1 PEDOT:PSS 0.34 0.06-945 8-888 1 × 10 at 300 K [70,77,78]
-2
2 PEDOT:Tos 0.37 6 × 10 -300 40-700 0.25 at RT [20]
-7 -2
3 PANI 0.02-0.542 10 -320 -16-225 1.1 × 10 at 423K [79-82]
-2
-2
4 PPy 0.2 6 × 10 -300 -1-40 3 × 10 at 423K [68,83]
-2 -3 -2
5 PTH 0.028-0.17 10 -10 10-100 2.9 × 10 at 250 K [84-86]
-5 -2
6 PC 0.34 4 × 10 -5 × 10 4.9-600 [76,87]
PEDOT:PSS: Poly(3,4-ethylenedioxythiophene): poly(styrenesulfonate); PANI: polyaniline; PPy: polypyrrole; PTH: polythiophene; PC:
polycarbazoles; PEDOT:Tos: poly(3,4-ethylenedioxythiophene): tosylate.
efficient charge transport .
[96]
In 2020, Liu et al. investigated incorporation thiophene unit in the structure of PDPP-4T and synthesized
PDPPT-5T and further modified the structure by replacing one thiophene unit with EDOT yielding
PDPPT-4T-EDOT. From the study, it has been ascertained that PDPP-4T-EDOT can attain comparatively
higher values of the Highest Occupied Molecular Orbital (HOMO), which facilitates efficient p-doping.
Theoretical calculations indicate that PDPP-4T-EDOT exhibits slightly improved coplanarity compared to
PDPP-5T to enhance charge carrier transport. Both polymers exhibit an increase in σ with an initial increase
in dopant concentration, but the σ decreases at higher concentrations and with it, the S also reduces. The PF
that the best results obtained by PDPP-4T-EDOT at a doping concentration of 0.5 mM was
-2
-2
-1
-1
298.2 μW m K , which is significantly superior to the value of 11.1 μW m K for PDPP-5T at 6 mM, as
PDPP-4T-EDOT gets easily doped due to its higher HOMO energy level. This work serves as proof
that engineering the energy levels by the incorporation of EDOT is beneficial for the developing
high-performance p-type thermoelectric polymers .
[97]
Similarly earlier on, selenophene substitution was demonstrated to be an effective approach for improving
intermolecular interactions and achieving high charge carrier mobility [98-100] . Ding et al. designed and
synthesized selenium-substituted DPP-based polymer for TE devices. They modified the PDPP-3T structure
by introducing a branched side chain on one side and a linear alkyl chain on the other end of each DPP
unit, resulting in PDPPS-12. The structure was further modified by replacing one sulfur atom with selenium
to obtain PDPPSe-12. The incorporation of selenium leads to strong intermolecular interactions and
ordered molecular packing. PDPPSe-12, a DPP-selenophene copolymer, displayed impressive hole mobility,
approaching 7 cm V s . Its maximal σ, when doped with FeCl , is nearly 997 S cm , over three times that
-1
-1 -1
2
3
of the doped PDPPS-12. Stronger intermolecular interactions occur within PDPPSe-12 because of larger
atomic radius of selenium compared to that of sulfur. As a result, PDPPSe-12’s molecular arrangement
remains mainly unaltered by doping, although PDPPS-12’s structure becomes less organized under similar
doping conditions. Atomic force microscopy (AFM) analysis revealed that FeCl doping profoundly altered
3
the morphology of PDPPS-12 by demolishing its fiber-like polycrystalline microstructure and reducing the
surface roughness. On the other hand, PDPPSe-12 retained its fiber-like intercalating network and exhibited
negligible morphological alteration upon doping. This suggests better dopant accommodation in
PDPPSe-12 with maintenance of its microstructure and facilitation of stable charge transport.
2
-1
Notably, at low doping levels, PDPPSe-12 exhibits a remarkable hole mobility of around 1.9 cm V s ,
-1
-2
leading to a maximum PF of 364 μW m K , which contributes to a ZT value of 0.25-marking a record for
-1
CP. This work provides insights into the structure-property relationship of doped OSc and indicates a
pathway for the rational design of high-performance OTE materials .
[101]