Page 195 - Read Online
P. 195
Page 12 of 18 Seo et al. Energy Mater. 2025, 5, 500123 https://dx.doi.org/10.20517/energymater.2025.38
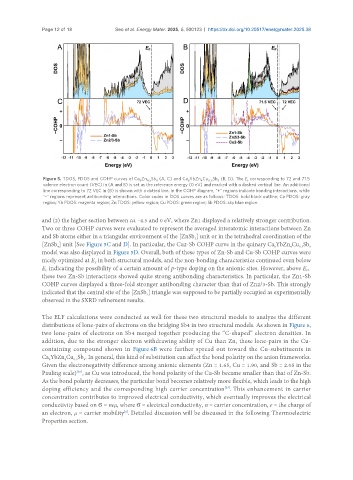
Figure 5. TDOS, PDOS and COHP curves of Ca Zn Sb (A, C) and Ca YbZn Cu Sb (B, D). The E corresponding to 72 and 71.5
9 4.5 9 8 4 0.5 9 F
valence electron count (VEC) in (A and B) is set as the reference energy (0 eV) and marked with a dashed vertical line. An additional
line corresponding to 72 VEC in (B) is shown with a dotted line. In the COHP diagram, "+" regions indicate bonding interactions, while
"-" regions represent antibonding interactions. Color codes in DOS curves are as follows: TDOS: bold black outline; Ca PDOS: gray
region; Yb PDOS: magenta region; Zn TDOS: yellow region; Cu PDOS: green region; Sb PDOS: sky blue region.
and (2) the higher section between ca. -4.5 and 0 eV, where Zn1 displayed a relatively stronger contribution.
Two or three COHP curves were evaluated to represent the averaged interatomic interactions between Zn
and Sb atoms either in a triangular environment of the [ZnSb ] unit or in the tetrahedral coordination of the
3
[ZnSb ] unit [See Figure 5C and D]. In particular, the Cu2-Sb COHP curve in the quinary Ca YbZn Cu Sb
9
8
4
0.5
4
model was also displayed in Figure 5D. Overall, both of these types of Zn-Sb and Cu-Sb COHP curves were
nicely optimized at E in both structural models, and the non-bonding characteristics continued even below
F
E indicating the possibility of a certain amount of p-type doping on the anionic sites. However, above E ,
F
F
these two Zn-Sb interactions showed quite strong antibonding characteristics. In particular, the Zn1-Sb
COHP curves displayed a three-fold stronger antibonding character than that of Zn2/3-Sb. This strongly
indicated that the central site of the [ZnSb ] triangle was supposed to be partially occupied as experimentally
3
observed in the SXRD refinement results.
The ELF calculations were conducted as well for these two structural models to analyze the different
distributions of lone-pairs of electrons on the bridging Sb4 in two structural models. As shown in Figure 6,
two lone-pairs of electrons on Sb4 merged together producing the “C-shaped” electron densities. In
addition, due to the stronger electron withdrawing ability of Cu than Zn, these lone-pairs in the Cu-
containing compound shown in Figure 6B were further spread out toward the Cu-substituents in
Ca YbZn Cu Sb . In general, this kind of substitution can affect the bond polarity on the anion frameworks.
9
0.5
8
4
Given the electronegativity difference among anionic elements (Zn = 1.65, Cu = 1.90, and Sb = 2.05 in the
Pauling scale) , as Cu was introduced, the bond polarity of the Cu-Sb became smaller than that of Zn-Sb.
[36]
As the bond polarity decreases, the particular bond becomes relatively more flexible, which leads to the high
doping efficiency and the corresponding high carrier concentration . This enhancement in carrier
[37]
concentration contributes to improved electrical conductivity, which eventually improves the electrical
conductivity based on σ = neμ, where σ = electrical conductivity, n = carrier concentration, e = the charge of
an electron, μ = carrier mobility . Detailed discussion will be discussed in the following Thermoelectric
[6]
Properties section.