Page 191 - Read Online
P. 191
Page 8 of 18 Seo et al. Energy Mater. 2025, 5, 500123 https://dx.doi.org/10.20517/energymater.2025.38
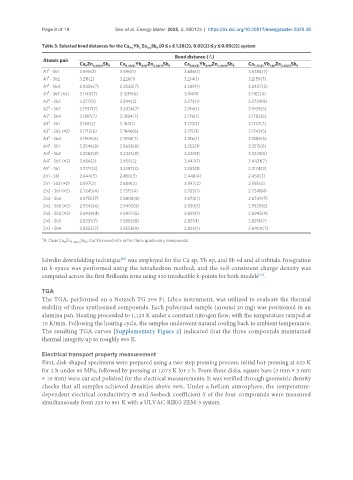
Table 3. Selected bond distances for the Ca Yb Zn Sb [0 ≤ x ≤ 1.28(2), 0.02(2) ≤ y ≤ 0.05(2)] system
x
9-x
4-y
9
Bond distance (Å)
Atomic pair
Ca Zn 4.48(2) Sb 9 Ca 8.43(2) Yb 0.57 Zn 4.46(1) Sb 9 Ca 8.01(3) Yb 0.99 Zn 4.46(1) Sb 9 Ca 7.72(2) Yb 1.28 Zn 4.45(2) Sb 9
9
a
A1 - Sb1 3.695(2) 3.696(1) 3.686(1) 3.6784(7)
a
A1 - Sb2 3.211(2) 3.226(1) 3.214(1) 3.2159(7)
a
A1 - Sb4 3.2426(7) 3.2543(7) 3.239(1) 3.2407(5)
a
A1 - Sb5 (×2) 3.1140(7) 3.1239(6) 3.114(1) 3.1162(4)
a
A2 - Sb2 3.377(2) 3.394(2) 3.373(1) 3.3729(8)
a
A2 - Sb3 3.1937(7) 3.2036(7) 3.194(1) 3.1939(5)
a
A2 - Sb4 3.1817(7) 3.1884(7) 3.176(1) 3.1782(5)
a
A3 - Sb1 3.169(2) 3.184(1) 3.170(1) 3.1737(7)
a
A3 - Sb3 (×2) 3.1712(8) 3.1846(6) 3.175(1) 3.1743(5)
a
A3 - Sb4 3.1909(8) 3.1998(7) 3.186(1) 3.1889(5)
a
A4 - Sb1 3.3546(8) 3.3638(8) 3.353(1) 3.3515(6)
a
A4 - Sb2 3.2282(8) 3.2435(8) 3.229(1) 3.2329(5)
a
A4 - Sb5 (×2) 3.454(2) 3.459(2) 3.447(1) 3.4421(7)
a
A5 - Sb1 3.3177(3) 3.3287(3) 3.353(1) 3.3174(3)
Zn1 - Sb1 2.444(3) 2.450(3) 2.446(4) 2.450(3)
Zn1 - Sb3 (×2) 2.597(2) 2.608(2) 2.597(2) 2.595(2)
Zn2 - Sb1 (×2) 2.7245(4) 2.7315(4) 2.723(1) 2.7241(4)
Zn2 - Sb4 2.6755(7) 2.6808(8) 2.674(1) 2.6749(7)
Zn2 - Sb5 (×2) 2.9243(6) 2.9492(8) 2.930(1) 2.9329(6)
Zn3 - Sb2 (×2) 2.6939(4) 2.6997(5) 2.693(1) 2.6945(4)
Zn3 - Sb3 2.8231(7) 2.8356(8) 2.821(1) 2.8218(7)
Zn3 - Sb4 2.8352(7) 2.8558(9) 2.838(1) 2.8400(7)
a
A: Ca in Ca Zn Sb ; Ca/Yb mixed-site in the three quaternary compounds.
9 4.48(2) 9
Löwdin downfolding technique was employed for the Ca 4p, Yb 6p, and Sb 5d and 4f orbitals. Integration
[29]
in k-space was performed using the tetrahedron method, and the self-consistent charge density was
computed across the first Brillouin zone using 320 irreducible k-points for both models .
[30]
TGA
The TGA, performed on a Netzsch TG 209 F1 Libra instrument, was utilized to evaluate the thermal
stability of three synthesized compounds. Each pulverized sample (around 20 mg) was positioned in an
alumina pan. Heating proceeded to 1,123 K under a constant nitrogen flow, with the temperature ramped at
10 K/min. Following the heating cycle, the samples underwent natural cooling back to ambient temperature.
The resulting TGA curves [Supplementary Figure 2] indicated that the three compounds maintained
thermal integrity up to roughly 900 K.
Electrical transport property measurement
First, disk-shaped specimens were prepared using a two-step pressing process: initial hot-pressing at 623 K
for 2 h under 60 MPa, followed by pressing at 1,073 K for 2 h. From these disks, square bars (3 mm × 3 mm
× 10 mm) were cut and polished for the electrical measurements. It was verified through geometric density
checks that all samples achieved densities above 96%. Under a helium atmosphere, the temperature-
dependent electrical conductivity σ and Seebeck coefficient S of the four compounds were measured
simultaneously from 323 to 861 K with a ULVAC-RIKO ZEM-3 system.